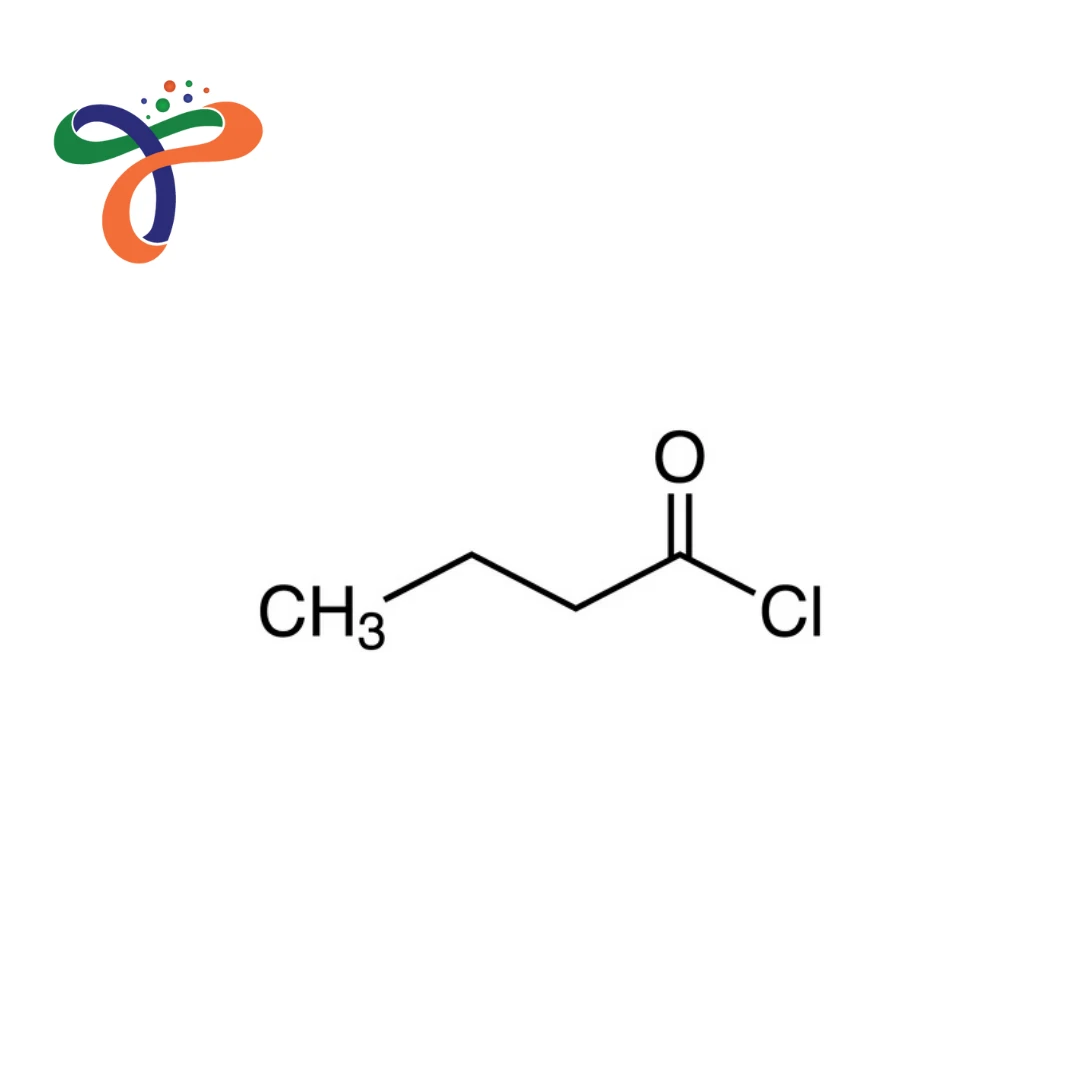
Butyryl Chloride | 141-75-3
Cas No :
141-75-3
Synonyms :
Form :
Liquid
Molecular Weight :
106.55 G/Mol
Molecular Formula :
C4H7CLO
Boiling Point :
78 °C
Melting Point :
-18 °C
Flash Point :
-5 °C
Solubility :
Reacts With Water; Soluble In Organic Solvents (Diethyl Ether, Chloroform, Benzene)
Description :
Overview of Butyryl Chloride
Butyryl Chloride, also known as Butanoyl Chloride, is a highly reactive industrial acid chloride compound widely used in chemical manufacturing. It is typically supplied as a clear, colourless to pale yellow liquid with a pungent odour and is valued for its acyl chloride (-COCl) functional group, enabling efficient acylation reactions in controlled industrial systems.
As an industrial chemical, Butyryl Chloride industrial grade is extensively used in organic synthesis, intermediate manufacturing, polymer modification, and specialty industrial formulations, where consistent purity and controlled reactivity are essential.
Applications of Butyryl Chloride
Chemical Manufacturing & Industrial Synthesis
Butyryl Chloride is widely used as an acylating agent in industrial organic synthesis, supporting esterification, amidation, and condensation reactions. It is commonly processed alongside Acetyl Chloride, Propionyl Chloride, and Thionyl Chloride in controlled acid chloride reaction systems.
Specialty Intermediates & Functional Chemical Production
In specialty chemical manufacturing, butyryl chloride is a key intermediate for producing performance-driven compounds. It is often evaluated with Benzoyl Chloride and Cyclopropanecarbonyl Chloride in advanced industrial transformation processes.
Polymer & Advanced Industrial Formulations
Butyryl Chloride is applied in polymer modification, coatings, and specialty formulations, where precise acylation improves structural properties and chemical stability under industrial processing conditions.
Safety & Handling Guidelines
- Keep it in a cool, dry, and well-ventilated location
- Be sure to protect yourself from humidity and moisture
- Seal containers well and keep them clearly labelled
- Avoid inhalation of vapours and contact with skin or eyes
- Use protective gloves, goggles, face shield, and suitable industrial clothing
- Handle under controlled industrial conditions
- Dispose of waste and packaging according to regulatory guidelines
Where to Buy Butyryl Chloride?
Butyryl Chloride Manufacturer
Butyryl Chloride is manufactured for industrial-grade applications requiring controlled purity, consistent acyl chloride content, and reliable performance in chemical manufacturing industries.
Butyryl Chloride Supplier & Distributor
Butyryl Chloride is available in large quantities in industrial and bulk packaging for companies that require dependable quality and controlled handling. Purchase Butyryl Chloride at Chemicalbull Pvt. Ltd. is a reputable industrial chemicals distributor and supplier that provides clients with dependable supply, documentation and safe logistics throughout India.
MSDS for Butyryl Chloride
The MSDS for Butyryl Chloride provides detailed information on hazards, safe handling practices, storage conditions, exposure controls, first-aid measures, and emergency response procedures. Always review the MSDS before industrial use.
Frequently Asked Questions (FAQs)
-
What is the chemical formula of Butyryl Chloride?
The chemical formula of Butyryl Chloride is C₄H₇ClO. -
What is the structure of Butyryl Chloride?
Butyryl Chloride contains a four-carbon alkyl chain attached to an acyl chloride (-COCl) functional group, enabling high reactivity in acylation systems. -
What is the common name of Butanoyl Chloride?
The common name of Butanoyl Chloride is Butyryl Chloride. -
What is Butyryl Chloride used for?
Butyryl Chloride is used in industrial organic synthesis, acylation reactions, intermediate production, polymer modification, and specialty chemical manufacturing. -
What is the difference between Butyryl Chloride and Acetyl Chloride?
Butyryl Chloride contains a four-carbon chain, while Acetyl Chloride contains a two-carbon chain, resulting in different reactivity and application profiles in industrial synthesis.