4-Aminophenol | 123-30-8
Cas No :
123-30-8
Synonyms :
Form :
Powder
Molecular Weight :
109.13 G/Mol
Molecular Formula :
C6H7NO
Boiling Point :
300 °C
Melting Point :
188 °C
Flash Point :
170 °C
Solubility :
Soluble In Water; Soluble In Ethanol; Sparingly Soluble In Diethyl Ether
Description :
Overview of 4-Aminophenol
4-Aminophenol is a vital industrial aromatic amine that is extensively utilised for its role as an intermediate in the chemical manufacturing process. It is usually sold as a white to mild brown crystalline substance, which can darken when exposed to air because of the process of oxidation. It is renowned due to its reactive amine and functional groups that are hydroxyl. This allows for an incredibly broad application in industrial synthesisers.
As an industrial chemical, 4-Aminophenol is used in dye intermediates, photographic chemicals, polymer additives, speciality chemical manufacturing, and advanced organic synthesis, where controlled reactivity and consistent quality are essential.
Applications of 4-Aminophenol
Chemical Manufacturing & Industrial Synthesis
4-Aminophenol is extensively used as an intermediate in industrial organic synthesis, supporting substitution, coupling, and functional transformation reactions. It is commonly processed alongside Nitrobenzene, 4-Nitroaniline, and 2-Aminopyridine in controlled aromatic chemical systems.
Dyes, Pigments & Photographic Chemicals
In industrial dye and photographic chemical production, 4-Aminophenol contributes to colour development and stabilisation processes. It is often evaluated with 1-Naphthol and Hydroxylamine Hydrochloride in specialised formulation systems.
Polymer Additives & Speciality Industrial Formulations
4-Aminophenol is applied in polymer modification and speciality chemical formulations, where its functional groups enhance stability, adhesion, and performance in demanding industrial applications.
Safety & Handling Guidelines
- Store in a cool, dry, and well-ventilated area
- Protect from air exposure to minimise oxidation
- Keep containers tightly sealed and clearly labelled
- Avoid inhalation of dust and contact with skin or eyes
- Use protective gloves, goggles, a face shield, and suitable industrial clothing
- Handle under controlled industrial conditions
- Dispose of waste and packaging according to regulatory guidelines
Where to Buy 4-Aminophenol?
4-Aminophenol Manufacturer
4-Aminophenol is manufactured for industrial-grade applications requiring controlled purity, stable composition, and reliable performance in dye and speciality chemical industries.
4-Aminophenol Supplier & Distributor
4-Aminophenol is offered in large quantities and in industrial packaging formats for companies that require dependable quality and controlled handling. Purchase 4-Aminophenol from Chemicalbull Pvt. Ltd., a reputable industrial chemicals distributor and supplier providing customers with dependable sourcing, documentation, and a compliant supply chain across India.
MSDS for 4-Aminophenol
The MSDS for 4-Aminophenol provides detailed information on hazards, safe handling practices, storage conditions, exposure controls, first-aid measures, and emergency response procedures. Always review the MSDS before industrial use.
Frequently Asked Questions (FAQs)
-
What is 4-Aminophenol used for?
4-Aminophenol can be found in the industrial process of chemical synthesis, dye manufacturing, photochemicals, polymer additives, and special formulas. -
Is Tylenol a 4-Aminophenol derivative?
Yes, acetaminophen (paracetamol) is chemically derived from 4-aminophenol, but 4-aminophenol itself is used primarily as an industrial intermediate. -
What is another name for 4-Aminophenol?
Another name for 4-Aminophenol is p-Aminophenol or para-aminophenol. -
What is the colour of 4-Aminophenol?
4-Aminophenol is typically white to light brown and may darken upon exposure to air due to oxidation. -
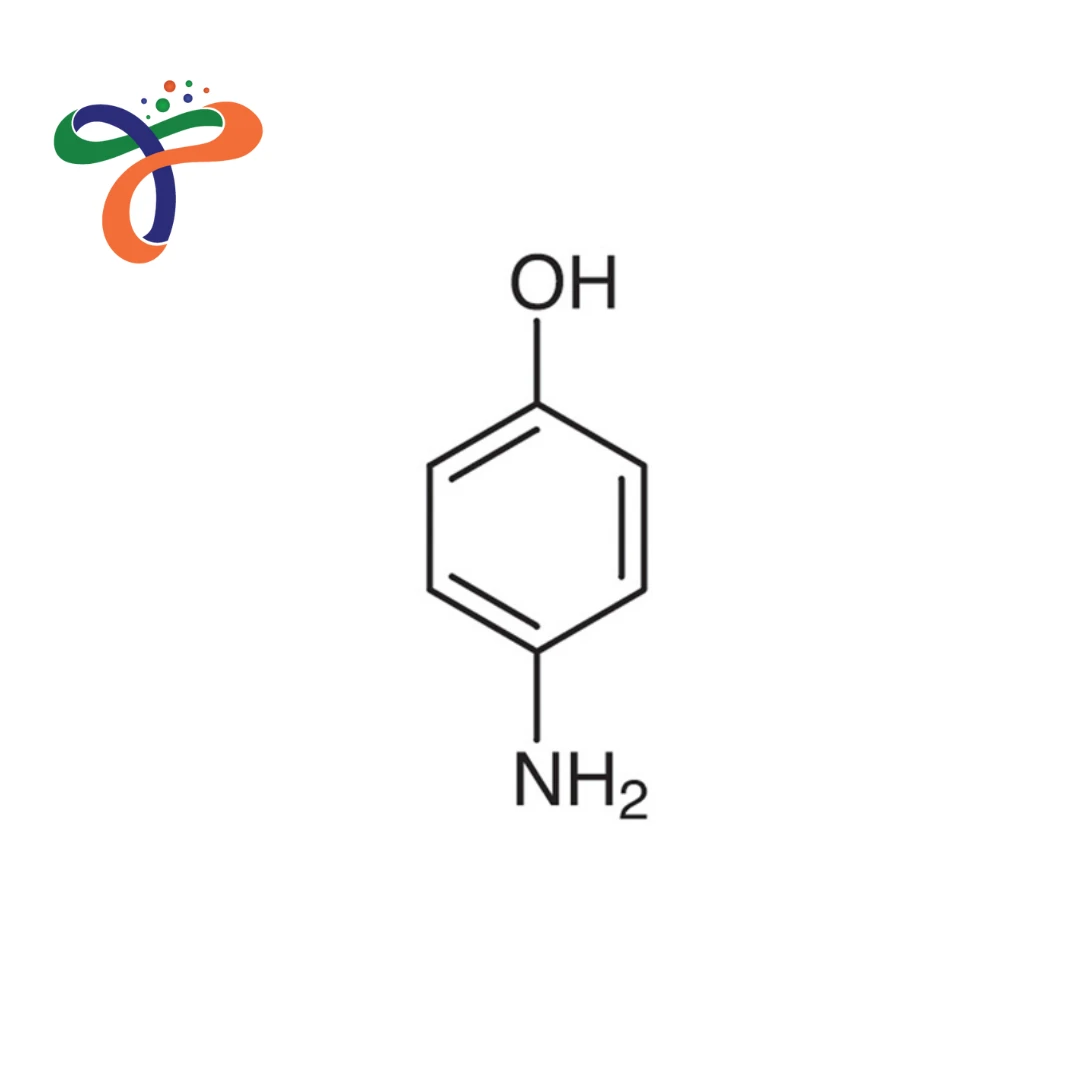
What is the structure of 4-Aminophenol?
4-Aminophenol consists of a benzene ring substituted with an amino group (-NH₂) and a hydroxyl group (-OH) at the para position, enabling multifunctional reactivity.