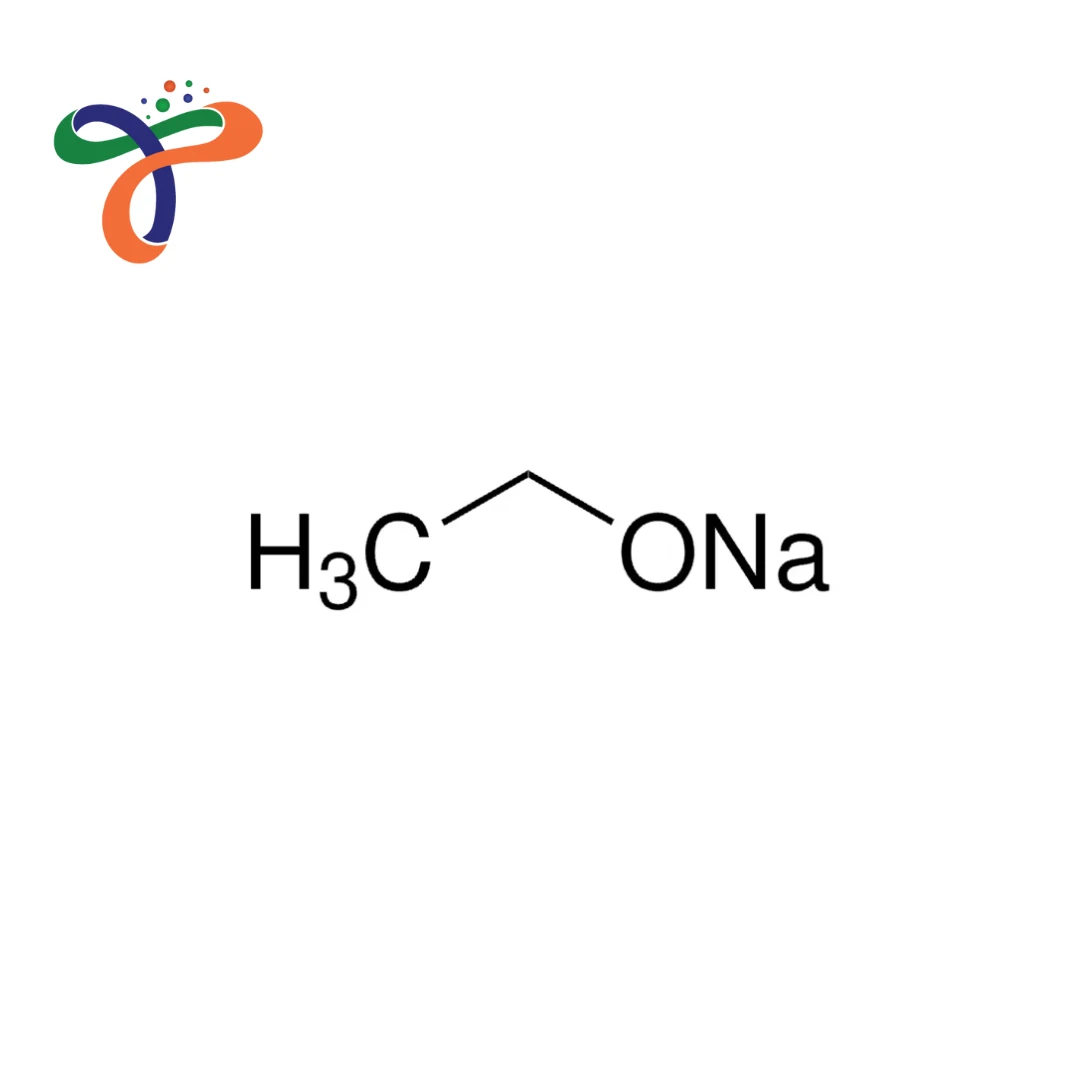
Sodium Ethoxide | 141-52-6
Cas No :
141-52-6
Synonyms :
Form :
Solid
Molecular Weight :
68.05 G/Mol
Molecular Formula :
C2H5NAO
Boiling Point :
200 °C
Melting Point :
157 °C
Flash Point :
12 °C
Solubility :
Soluble In Ethanol; Reacts With Water To Form Ethanol And Sodium Hydroxide
Description :
Overview of Sodium Ethoxide
Sodium Ethoxide, also known as Sodium Ethylolate, is a potent alkoxide and a strong nucleophilic base with a Molecular Weight of 68.05 g/mol and a Melting Point of 260°C (Decomposes). Sodium Ethoxide Chemical Formula(structure) is C2H5NaO. As a premier Industrial Chemical Supplier, Chemical Bull manufactures and distributes Sodium Ethoxide in both a free-flowing 98% powder and a 21% ethanol solution.
Identified by CAS 141-52-6, this compound is significantly more basic than sodium hydroxide. It is a cornerstone reagent in anhydrous organic chemistry, favored for its ability to facilitate complex carbon-carbon bond formations without the interference of water.
Application of Sodium Ethoxide
The Sodium Ethoxide manufacturing process at Chemical Bull involves the controlled reaction of high-purity sodium metal with anhydrous ethanol under a strict nitrogen blanket. This prevents the formation of sodium hydroxide and ensures the active content remains stable.
Pharmaceutical Synthesis
In the Pharmaceutical Intermediates, it is a "Star" reagent:
-
Condensation Reactions: It is the primary catalyst for Claisen Condensations, used to synthesize esters, vitamins (such as Vitamin B1), and various barbiturates.
-
Ring Closure: Facilitates the formation of heterocyclic rings essential for many Active Pharmaceutical Ingredients (APIs).
-
Synergistic Synthesis: It is frequently used alongside reagents like triphosgene in advanced organic synthesis to build complex molecular frameworks for modern medicine.
Biodiesel Manufacture
Sodium Ethoxide is the preferred industrial use catalyst in high-efficiency energy plants:
-
Transesterification: It converts triglycerides into biodiesel more effectively than traditional hydroxides. Because it does not produce water as a byproduct, it prevents "soaping" and maximizes fuel yield. During final processing, organic acids like Lactic Acid may be used for precise pH neutralization.
Fine Chemical & Organic Synthesis
-
Ether Synthesis: A key nucleophile in Williamson Ether Synthesis for the production of high-end fragrances and dyes.
-
Solvent Compatibility: Often formulated with anhydrous solvents like Ethyl Lactate to ensure reaction stability in moisture-sensitive environments
Safety, Handling & Professional Storage
Due to its high reactivity and corrosive nature, safe handling is mandatory:
-
Handling: Sodium Ethoxide is highly alkaline and moisture-sensitive. It reacts with atmospheric humidity to form Sodium Hydroxide and Ethanol. Always use specialized PPE, including nitrile gloves and a full-face shield, to prevent chemical burns.
-
Storage: Store in a cool, dry, and nitrogen-purged environment. To maintain chemical integrity, containers must be hermetically sealed. This prevents the product from absorbing CO_2 and degrading into sodium carbonate.
-
Regulatory Compliance: Every batch is rigorously tested for "Free Alkali" and "Active Content," backed by a comprehensive COA and MSDS.
Why Buy from Chemical Bull?
Sodium Ethoxide Manufacturer
-
Assay Stability: We guarantee high-purity levels by using vacuum-sealed, moisture-barrier packaging that protects the product's reactive potency.
-
Bulk Versatility: We support manufacturing requirements ranging from small-scale R&D to metric-ton industrial production.
-
Technical Support: Our team provides the exact solubility and purity profiles required to satisfy your internal Quality Assurance (QA) and safety protocols.
Sodium Ethoxide Supplier & Distributor
we cater to both small-scale R&D labs and large-scale industrial plants. We provide flexible packaging options, ranging from vacuum-sealed moisture-barrier bags for 98% powder to specialized steel drums for 21% liquid solutions, ensuring safe transit to any industrial hub.
MSDS Sodium Ethoxide
Due to its corrosive and moisture-sensitive nature, safe handling is our priority. Every batch is supplied with an updated MSDS (Material Safety Data Sheet) and a COA (Certificate of Analysis). These documents provide critical data on flammability, pH, and emergency neutralisation procedures to ensure your facility meets international safety standards.
Frequently Asked Questions (FAQs)
- Why is the 260°C Melting Point significant?
While it shows the solid's thermal limits, the product decomposes at this temperature. For manufacturing safety, reactions using Sodium Ethoxide are typically performed at much lower temperatures. - What is the shelf life of the 98% powder?
In original, nitrogen-blanketed packaging, it remains stable for 12 months. Exposure to air will cause rapid degradation through moisture absorption. - Can Sodium Ethoxide be used in water-based reactions?
Absolutely not. It reacts immediately and violently with water. All Sodium Ethoxide uses must be conducted in anhydrous (water-free) organic solvents.