Doxorubicin Hydrochloride | 25316-40-9
Cas No :
25316-40-9
Synonyms :
Form :
Powder
Molecular Weight :
579.98 G/Mol
Molecular Formula :
C27H30CLNO11
Boiling Point :
570 °C
Melting Point :
223 °C
Flash Point :
300 °C
Solubility :
Freely Soluble In Water; Soluble In Methanol; Insoluble In Nonpolar Solvents
Description :
Overview of Doxorubicin Hydrochloride
Doxorubicin Hydrochloride is a pharmaceutical-grade anthracycline antibiotic used as a high-potency oncology active pharmaceutical ingredient. It is supplied as the hydrochloride salt to enhance aqueous compatibility and formulation stability for regulated cancer therapies.
In pharmaceutical manufacturing, Doxorubicin Hydrochloride is valued for its broad antitumour activity, well-established clinical profile, and suitability for sterile dosage forms. It is produced under stringent GMP and containment conditions due to its cytotoxic nature.
As a pharma chemical, Doxorubicin Hydrochloride is used exclusively in oncology treatment formulations.
Applications of Doxorubicin Hydrochloride
Oncology Chemotherapy Formulations
Doxorubicin Hydrochloride is used as an active pharmaceutical ingredient in chemotherapy formulations for the treatment of multiple solid tumours and haematological malignancies. It is incorporated into treatment protocols where strong antineoplastic activity is required.
Combination Cancer Therapy Regimens
Doxorubicin Hydrochloride is widely used in combination chemotherapy regimens to enhance therapeutic effectiveness across different cancer types.
Regulated Oncology API Manufacturing
Doxorubicin Hydrochloride is manufactured as a cytotoxic pharmaceutical raw material with strict control over potency, impurity profile, and microbial limits. It is suitable for highly regulated oncology manufacturing environments with specialised safety controls.
In oncology API portfolios, it is often evaluated alongside Epirubicin Hydrochloride, Daunorubicin Hydrochloride, and Paclitaxel IP for therapy planning and formulation development.
Safety & Handling Guidelines
-
Store under refrigerated or controlled temperature conditions
-
Protect from light and moisture
-
Keep containers tightly sealed and clearly labelled
-
Avoid inhalation or direct contact during handling
-
Use cytotoxic-handling personal protective equipment
-
Handle only in controlled oncology manufacturing areas
-
Dispose of waste according to biomedical and regulatory guidelines
Where to Buy Doxorubicin Hydrochloride?
Doxorubicin Hydrochloride Manufacturer
Doxorubicin Hydrochloride is manufactured for pharmaceutical oncology applications where controlled synthesis, cytotoxic containment, and regulatory compliance are essential.
Doxorubicin Hydrochloride Supplier & Distributor
Pharmaceutical-grade Doxorubicin Hydrochloride is supplied in suitable packaging for licensed oncology formulation manufacturers. As a pharmaceutical chemicals supplier and distributor, Chemicalbull Pvt. Ltd. supports compliant sourcing with standard documentation.
MSDS for Doxorubicin Hydrochloride
The MSDS for Doxorubicin Hydrochloride provides detailed information on hazards, handling precautions, storage conditions, exposure controls, and emergency response measures. It must be reviewed before oncology API handling or formulation.
Frequently Asked Questions (FAQs)
-
What is Doxorubicin Hydrochloride used for?
Doxorubicin Hydrochloride is used in pharmaceutical oncology formulations for the treatment of various cancers. -
What is the solubility of Doxorubicin Hydrochloride?
Doxorubicin hydrochloride solubility is high in water, making it suitable for sterile injectable formulations. -
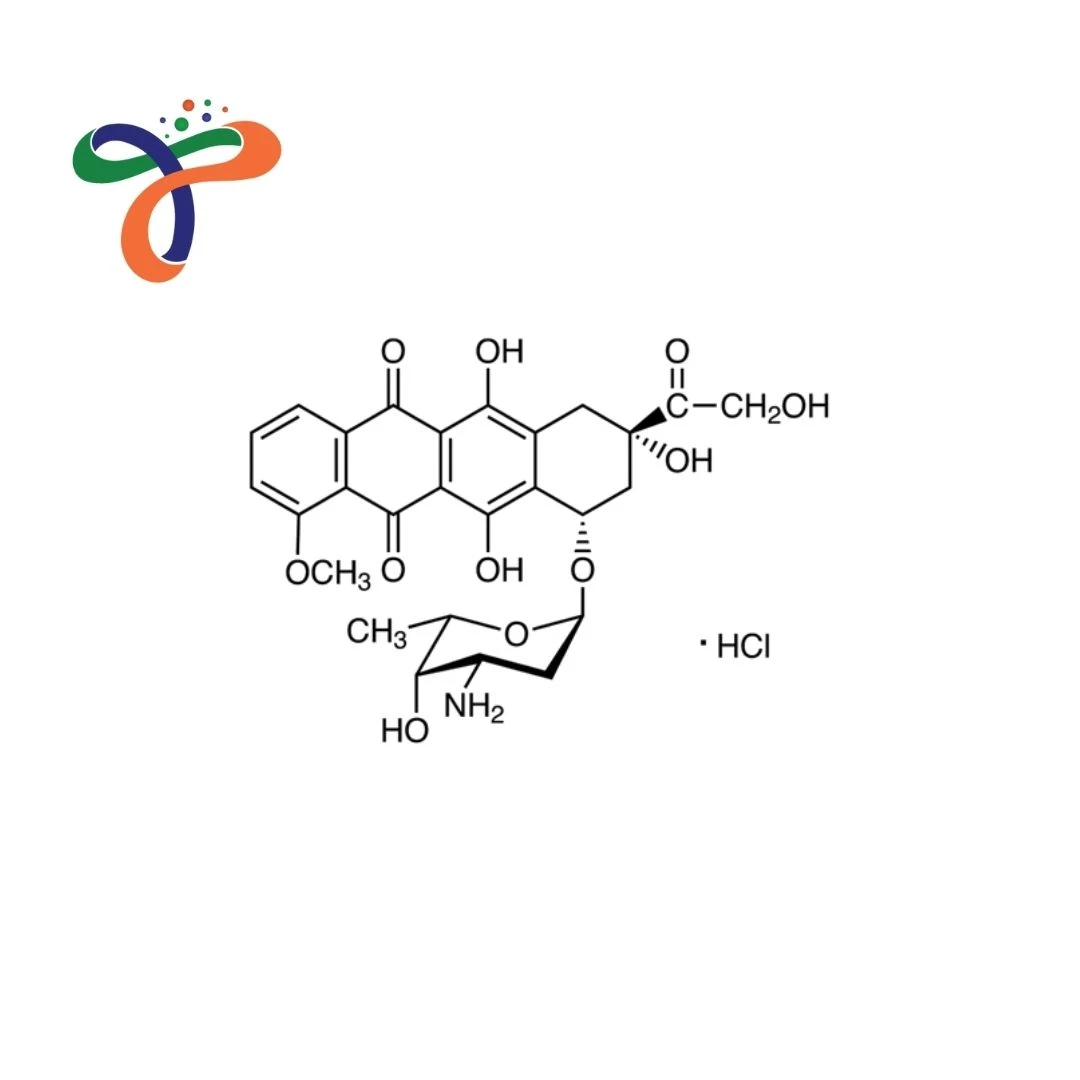
What is the structure of Doxorubicin Hydrochloride?
Doxorubicin hydrochloride structure consists of an anthracycline core supplied as a hydrochloride salt for pharmaceutical use. -
Is Doxorubicin Hydrochloride an API?
Yes, Doxorubicin Hydrochloride is a cytotoxic active pharmaceutical ingredient used in cancer therapy. -
Is Doxorubicin Hydrochloride suitable for regulated manufacturing?
Yes, it is produced under strict GMP and cytotoxic-handling regulations for oncology use.