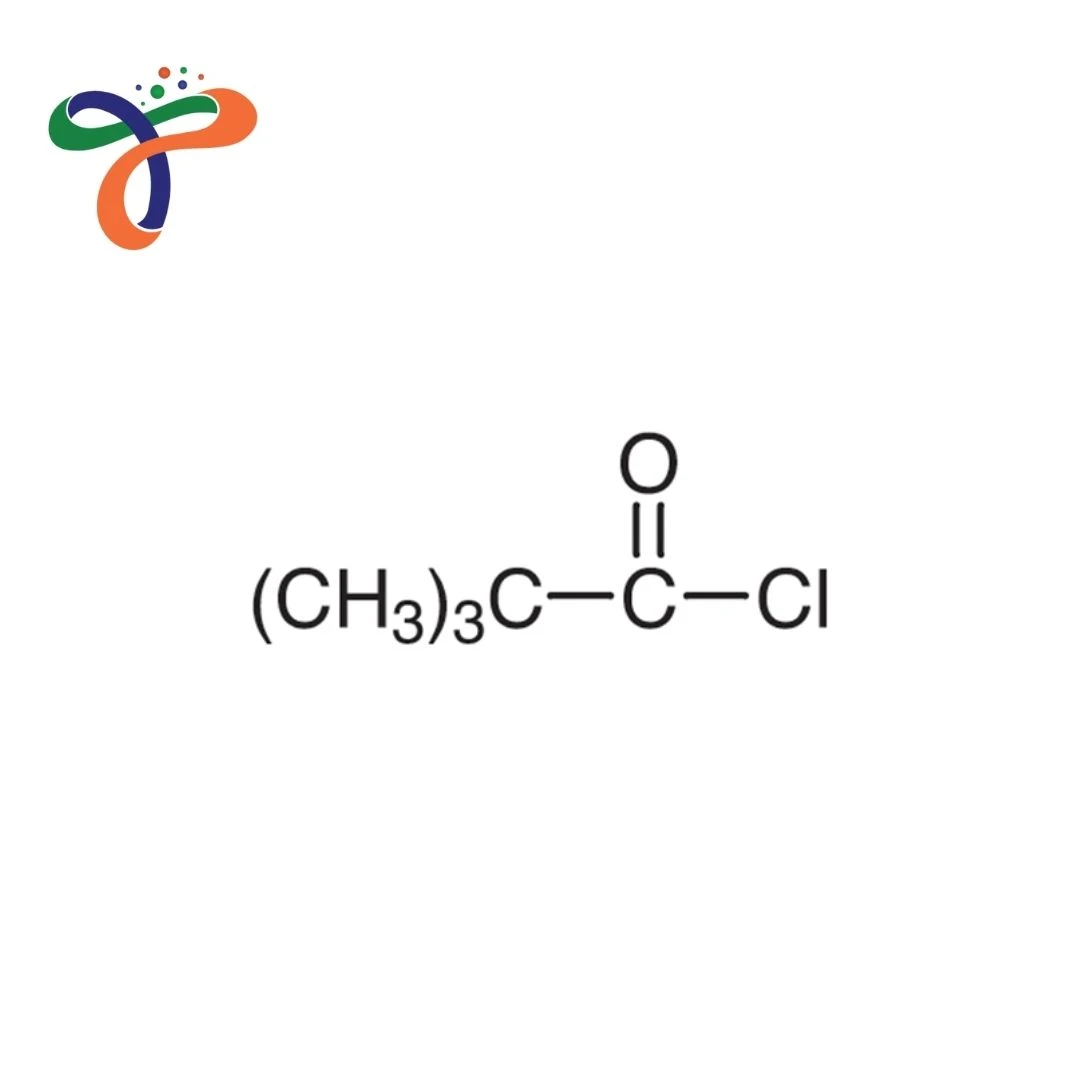
Trimethylacetyl Chloride | 3282-30-2
Cas No :
3282-30-2
Form :
Liquid
Molecular Weight :
120.57 G/Mol
Molecular Formula :
C5H9CLO
Boiling Point :
83 °C
Melting Point :
-60 °C
Flash Point :
-21 °C
Solubility :
Reacts With Water To Form Pivalic Acid And Hcl; Soluble In Organic Solvents; Insoluble In Water
Description :
Overview of Trimethylacetyl Chloride
Trimethylacetyl Chloride (Pivaloyl Chloride) is a highly reactive acyl chloride used as an acylating agent, pharmaceutical intermediate, and reagent in organic synthesis.
Known for its bulky tert-butyl group, which provides steric hindrance and resistance to hydrolysis, Trimethylacetyl Chloride is widely used in pharmaceutical manufacturing, protecting group chemistry, and the preparation of pivaloyl derivatives.
This compound appears as a colorless to pale yellow liquid with a characteristic pungent, acrid odor. Its neopentyl acid chloride structure provides unique reactivity patterns that are useful for selective acylation.
Related products include Acetyl Chloride, Benzoyl Chloride, Trimethylsulfoxonium Iodide, and other acylating agents used in pharmaceutical synthesis.
Applications of Trimethylacetyl Chloride
Pharmaceutical Synthesis
- Used in introducing pivaloyl protecting groups
- Applied in synthesizing pharmaceutical intermediates
- Used in preparing pivaloyl esters (prodrugs)
- Applied in manufacturing antibiotics (pivampicillin, pivmecillinam)
Industrial Chemical Synthesis
- Used as an acylating agent in organic reactions
- Applied in preparing esters and amides
- Used in synthesizing specialty chemicals
- Applied in polymer modification
Protecting Group Chemistry
- Used in O-acylation of alcohols
- Applied in the N-acylation of amines
- Used in peptide synthesis
Safety & Handling Guidelines
- HIGHLY REACTIVE and CORROSIVE—handle with extreme care
- LACHRYMATORY—causes severe eye and respiratory irritation
- Reacts violently with water and moisture
- Use only in a well-ventilated fume hood
- Wear appropriate PPE, including acid-resistant gloves and a face shield
- Handle under strictly anhydrous conditions
Where to Buy Trimethylacetyl Chloride?
Trimethylacetyl Chloride Manufacturer
ChemicalBull supplies high-purity Trimethylacetyl Chloride for pharmaceutical and research applications.
Trimethylacetyl Chloride Supplier & Distributor
- Available in controlled quantities for pharmaceutical and research use
- High-purity anhydrous grade (98%, 99%)
- Special moisture-resistant packaging
- COA, MSDS, and comprehensive safety documentation provided
MSDS
Includes comprehensive reactivity warnings, corrosivity information, and strict handling protocols.
Frequently Asked Questions (FAQs)
-
What is Trimethylacetyl Chloride used for?
Used as an acylating agent in pharmaceutical synthesis, introducing pivaloyl protecting groups, preparing prodrugs, and manufacturing antibiotic intermediates. -
What is the molecular formula?
Molecular formula: C₅H₉ClO or (CH₃)₃CCOCl with molecular weight 120.58 g/mol. -
Why is it called Pivaloyl Chloride?
Derived from pivalic acid (trimethylacetic acid), the "pival-" prefix indicates the trimethyl-substituted structure.