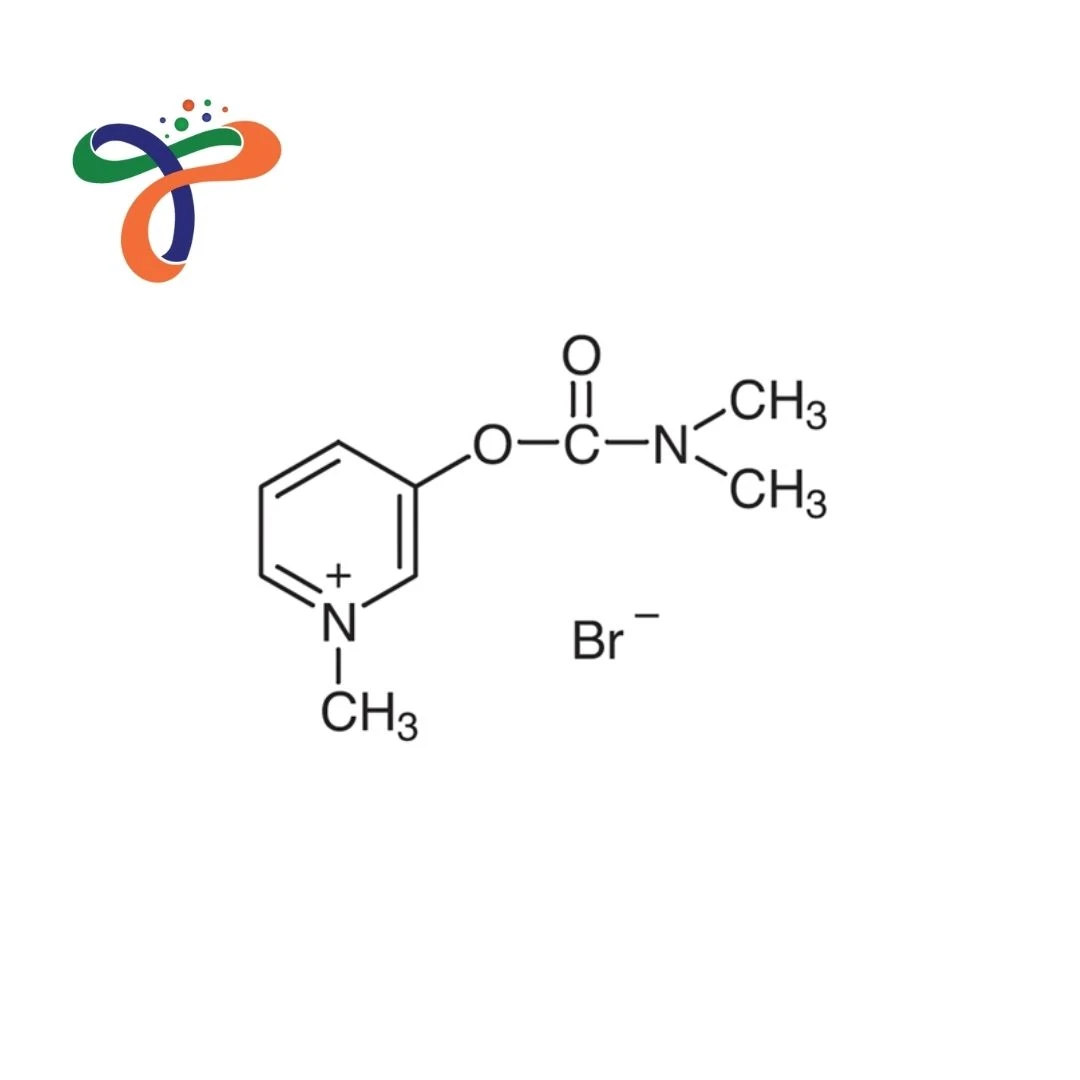
Pyridostigmine Bromide | 101-26-8
Cas No :
101-26-8
Synonyms :
Form :
Solid
Molecular Weight :
365.29 G/Mol
Molecular Formula :
C9H13BRN2O2
Boiling Point :
290 °C
Melting Point :
210 °C
Flash Point :
210 °C
Solubility :
Very Soluble In Water; Soluble In Methanol And Ethanol; Insoluble In Non-Polar Solvents
Description :
Overview of Pyridostigmine Bromide
Pyridostigmine Bromide is a cholinesterase inhibitor pharmaceutical active ingredient primarily used in the management of neuromuscular disorders, especially myasthenia gravis. It works by inhibiting acetylcholinesterase, thereby increasing the availability of acetylcholine at neuromuscular junctions and improving muscle strength.
Pyridostigmine Bromide appears as a white to off-white crystalline powder and is freely soluble in water. It exhibits good chemical stability and is suitable for oral pharmaceutical formulations such as tablets, syrups, and sustained-release dosage forms. Due to its well-established therapeutic profile, it is widely used in long-term neurological treatment regimens.
ChemicalBull supplies pharma-grade Pyridostigmine Bromide with high purity, consistent particle size, and reliable batch-to-batch quality suitable for regulated pharmaceutical manufacturing. Each shipment is supported with complete documentation, including MSDS, COA, and technical data sheets.
As a trusted chemical supplier and reliable distributor, ChemicalBull ensures dependable availability of Pyridostigmine Bromide. You may also like Neostigmine Methylsulfate, Donepezil Hydrochloride, Rivastigmine, and Pharmaceutical APIs for related neurological and cholinergic applications.
Applications of Pyridostigmine Bromide
Pharmaceutical Formulations
-
Used in tablets and oral liquid formulations
-
Applied in immediate and extended-release dosage forms
-
Suitable for long-term neurological therapy
Neurology & Clinical Use
-
Used in the treatment of myasthenia gravis
-
Applied to neuromuscular transmission disorders
-
Widely prescribed in neurological care
Hospital & Therapeutic Use
-
Used in chronic neuromuscular disease management
-
Applied in post-surgical and rehabilitation therapy
-
Accepted in global clinical practice
Pharmaceutical Research & Development
-
Used in bioequivalence and formulation studies
-
Applied in cholinergic system research
-
Suitable for controlled pharmaceutical laboratories
Safety & Handling Guidelines
-
Handle using gloves and appropriate protective equipment
-
Avoid inhalation of dust
-
Use in well-ventilated pharmaceutical manufacturing areas
-
Store in tightly sealed containers away from moisture
-
Follow MSDS and standard pharmaceutical safety practices
Where to Buy Pyridostigmine Bromide?
Pyridostigmine Bromide Manufacturer
ChemicalBull supplies pharmaceutical-grade Pyridostigmine Bromide suitable for neuromuscular and neurological drug formulations.
Pyridostigmine Bromide Supplier & Distributor
-
Bulk and customised pharmaceutical packaging options
-
Export-grade quality with COA, TDS & MSDS
-
Reliable supply across India and international markets
-
Trusted chemical supplier for pharmaceutical APIs
Pyridostigmine Bromide MSDS
The Pyridostigmine Bromide MSDS provides essential information on safe handling, storage conditions, exposure controls, stability, and emergency procedures. Always review the MSDS before bulk handling or pharmaceutical manufacturing use.
Frequently Asked Questions (FAQs)
-
Why is Pyridostigmine Bromide preferred for long-term therapy?
It has a predictable duration of action and is suitable for chronic management of neuromuscular disorders. -
Is Pyridostigmine Bromide water-soluble?
Yes, it is freely soluble in water, making it suitable for oral liquid formulations. -
Does Pyridostigmine Bromide act on the central nervous system?
Its primary action is peripheral, mainly at neuromuscular junctions. -
Is Pyridostigmine Bromide suitable for sustained-release formulations?
Yes, it is commonly formulated into extended-release tablets for controlled dosing. -
Is safety documentation provided with the supply?
Yes, every shipment includes MSDS, COA, and complete technical documentation.