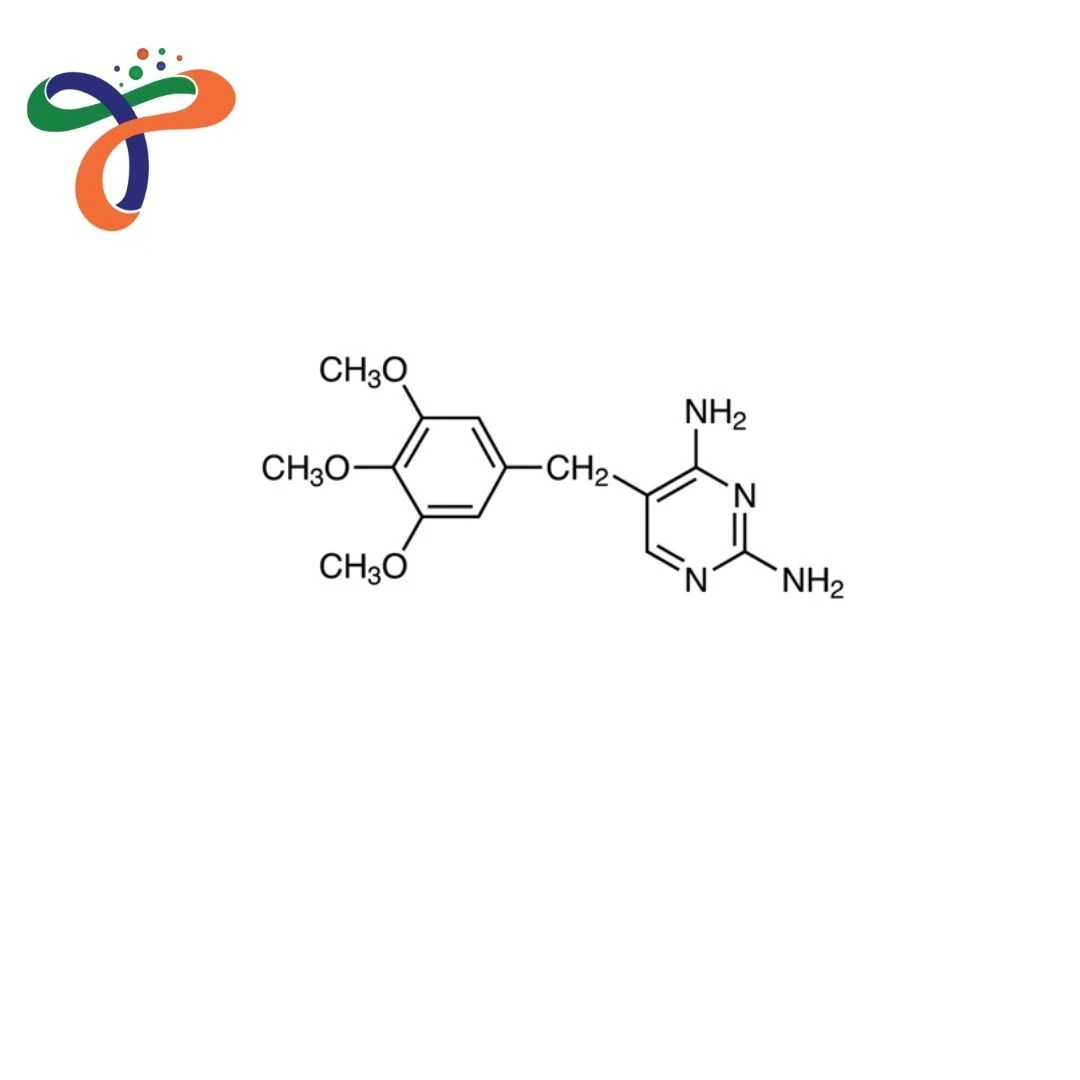
Trimethoprim | 738-70-5
Cas No :
738-70-5
Form :
Powder
Molecular Weight :
290.32 G/Mol
Molecular Formula :
C14H18N4O3
Boiling Point :
235 °C
Melting Point :
164 °C
Flash Point :
210 °C
Solubility :
Slightly Soluble In Water; Soluble In Methanol And Ethanol; Soluble In Dimethylformamide
Description :
Overview of Trimethoprim
Trimethoprim is a pharmaceutical-grade antibacterial drug widely employed as a pharmaceutical active ingredient in anti-infective formulations for systemic use. It is part of the diaminopyrimidine class and is frequently used in single-agent as well as combination antibiotics.
In pharmaceutical manufacturing, Trimethoprim is valued for its targeted antibacterial activity, formulation compatibility, and stable performance in oral dosage forms. It is produced under regulated GMP conditions to meet pharmacopeial standards for purity, assay, and impurity control.
As a pharmaceutical chemical, Trimethoprim plays a critical role in antibacterial therapy and combination drug development.
Applications of Trimethoprim
Antibacterial Formulation Development
Trimethoprim is widely used in oral tablets, capsules, and suspension formulations intended for the treatment of bacterial infections. It is particularly useful in urinary tract infection–focused products and other systemic antibacterial therapies.
Combination Antibiotic Manufacturing
Trimethoprim is often used together with sulfonamide antibacterial drugs to boost the effectiveness of antibacterial drugs. It is a synergistic antimicrobial agent in fixed-dose combination drugs that are developed in accordance with pharmaceutical standards.
API Synthesis & Process Development
Trimethoprim synthesis is carried out under controlled pharmaceutical manufacturing environments to ensure high purity and consistent batch quality. It is incorporated into scalable production pipelines for global antibiotic markets.
Regulated Pharmaceutical Production
Trimethoprim is produced as a highly pure pharmaceutical raw material, with strict control over solvents that remain impurities, along with stability factors. The material is ideal for GMP-compliant large-scale production of antibiotics.
In antibacterial product portfolios, it is often evaluated alongside Sulfamethoxazole, Ciprofloxacin Hydrochloride, and Doxycycline Hydrochloride for therapeutic planning and formulation optimisation.
Safety & Handling Guidelines
- Store in a cool, dry, and controlled environment
- Protect from moisture and excessive heat
- Keep containers tightly sealed and clearly labelled
- Avoid inhalation of powder during handling
- Make sure you have the appropriate personal protection equipment
- Handle under controlled manufacturing conditions
- The disposal of waste should be in line with the regulations
Where to Buy Trimethoprim?
Trimethoprim Manufacturer
Trimethoprim is manufactured for pharmaceutical applications where controlled potency, consistent quality, and regulatory compliance are essential.
Trimethoprim Supplier & Distributor
Pharmaceutical-grade Trimethoprim is supplied in suitable packaging for formulation manufacturers. As a pharmaceutical chemicals supplier and distributor, Chemicalbull Pvt. Ltd. supports reliable sourcing with standard documentation.
MSDS for Trimethoprim
The MSDS for Trimethoprim contains detailed information about the dangers, safe handling procedures, the storage environment, exposure control and emergency response protocols. It is recommended to review it prior to using the pharmaceutical manufacturing.
Frequently Asked Questions (FAQs)
-
What is Trimethoprim used for?
Trimethoprim is used in pharmaceutical formulations for the treatment of bacterial infections, particularly urinary tract infections. -
What is the mechanism of action of Trimethoprim?
The mechanism of action of trimethoprim involves inhibition of bacterial dihydrofolate reductase, which interferes with folic acid synthesis and bacterial growth. -
What is the chemical name for Trimethoprim?
The chemical name for Trimethoprim is 2,4-diamino-5-(3,4,5-trimethoxybenzyl)pyrimidine.