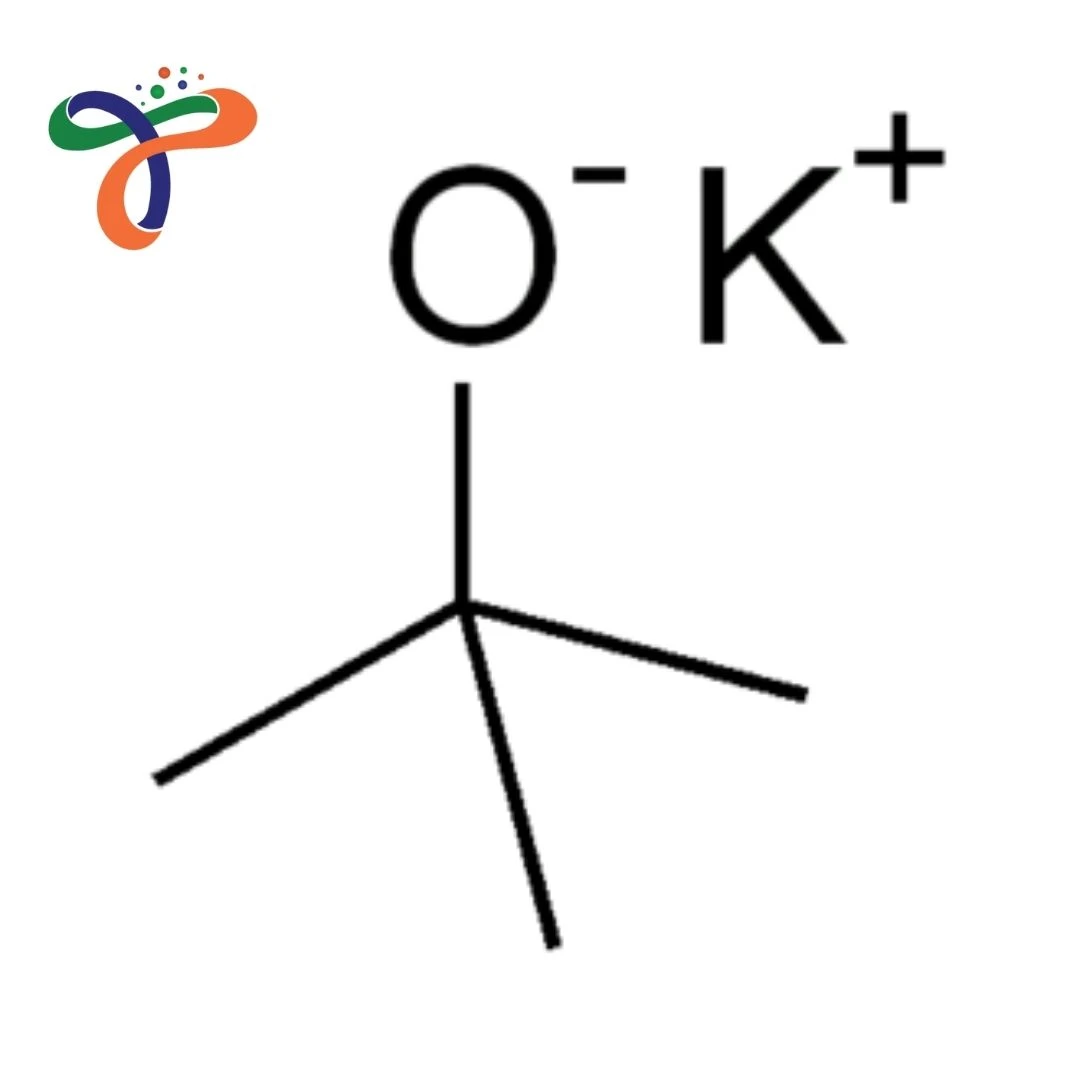
Potassium Tert-Butoxide | 865-47-4
Cas No :
865-47-4
Synonyms :
Form :
Solid
Molecular Weight :
112.21 G/Mol
Molecular Formula :
C4H9KO
Boiling Point :
200 °C
Melting Point :
72 °C
Flash Point :
240 °C
Solubility :
Insoluble In Water; Soluble In Diethyl Ether And Thf
Description :
Overview of Potassium Tert-Butoxide
Potassium Tert Butoxide (KOtBu) is very reactive, and a robust base widely used in chemical synthesis, manufacturing of pharmaceuticals, as well as a reagent in different transformations. It is renowned for being among the most robust non-nucleophilic base options as well as its superior removal reaction selectivity. Potassium Tert Butoxide is extensively used in Williamson's synthesis of ethers, E2 elimination, as well as many additional organic reactions.
This chemical appears as a pale to white powder, which is very sensitive to moisture. Its tert-butoxide-rich tert group is a bulky solid with basicity, while also reducing its nucleophilicity, which makes it an excellent choice in base-catalyzed reactions.
Other products related to this comprise Sodium Tert Butoxide Potassium Methoxide, Lithium Diisopropylamide (LDA), as well as other non-nucleophilic bases that are used for organic syntheses. It can be utilized to make reactions using Diethyl Maleate as well as Diethyl Oxalate, which are substrates.
Applications of Potassium Tert-Butoxide
Organic Synthesis
- Utilized for E2 removal reactions
- Used to Williamson synthesizing ether
- Utilized in condensing reactions (Claisen, Knoevenagel)
- Applied in preparing enolates
- It is used for aryl coupling reactions
- Utilized in the C-H functionalization
- It is used to synthesize pharmaceuticals
- Used in the preparation of heterocyclic compounds
Pharmaceutical Synthesis
- It is used in the manufacture of active components for pharmaceuticals.
- Utilized in preparing pharmaceutical intermediates
- It is used to synthesize potential drug compounds
- Used to research medicinal chemistry
- Utilized for complex molecule synthesis
- It is used in the production of fine chemicals
- Utilized in synthesis with a higher level of scale
- Used to process the optimization of chemistry
Advanced Organic Transformations
- used for cross-coupling reactions (Buchwald-Hartwig)
- Utilized in reactions of metalization
- It is used in the preparation of organic compounds
- Used to the polymer chemical industry
- It is used in materials science applications
- Applied in developing new synthetic methodologies
Safety & Handling Guidelines
- PYROPHORIC and EXTREMELY REACTIVE—ignites spontaneously with moisture
- Reacts violently with water, acids, and protic solvents
- Use only under strictly anhydrous conditions in an inert atmosphere
- Handle in the glove box or using Schlenk techniques
- Wear full PPE, including chemical-resistant gloves, face shield
- Store in tightly sealed containers under nitrogen or argon
- Keep away from all moisture sources, acids, and oxidizers
- Have a dry chemical fire extinguisher available (NEVER use water)
- Dispose of according to hazardous material protocols
- Follow COA and MSDS instructions strictly
Where to Buy Potassium Tert-Butoxide?
Potassium Tert-Butoxide Manufacturer
ChemicalBull supplies high-purity Potassium Tert-Butoxide for pharmaceutical and research applications with appropriate safety packaging.
Potassium Tert-Butoxide Supplier & Distributor
- Available as powder or solution in THF or toluene
- Multiple concentration options for solutions
- High-purity anhydrous grades
- Packaged under an inert atmosphere
- COA, MSDS, and comprehensive safety documentation provided
MSDS
Includes comprehensive pyrophoric material handling instructions, fire hazard warnings, and emergency procedures.
Frequently Asked Questions (FAQs)
-
What is Potassium Tert-Butoxide CAS No?
CAS number: 865-47-4. -
What is Potassium Tert-Butoxide's molecular weight?
Molecular weight: 112.21 g/mol. -
What is Potassium Tert-Butoxide formula?
Molecular formula: C₄H₉KO or (CH₃)₃COK. -
What is Potassium Tert-Butoxide solubility?
Soluble in THF, toluene, and other aprotic solvents; reacts with protic solvents, including alcohols (except tert-butanol). -
What is Potassium Tert-Butoxide used for?
Used as a strong non-nucleophilic base in elimination reactions, ether synthesis, condensations, pharmaceutical synthesis, and various organic transformations.