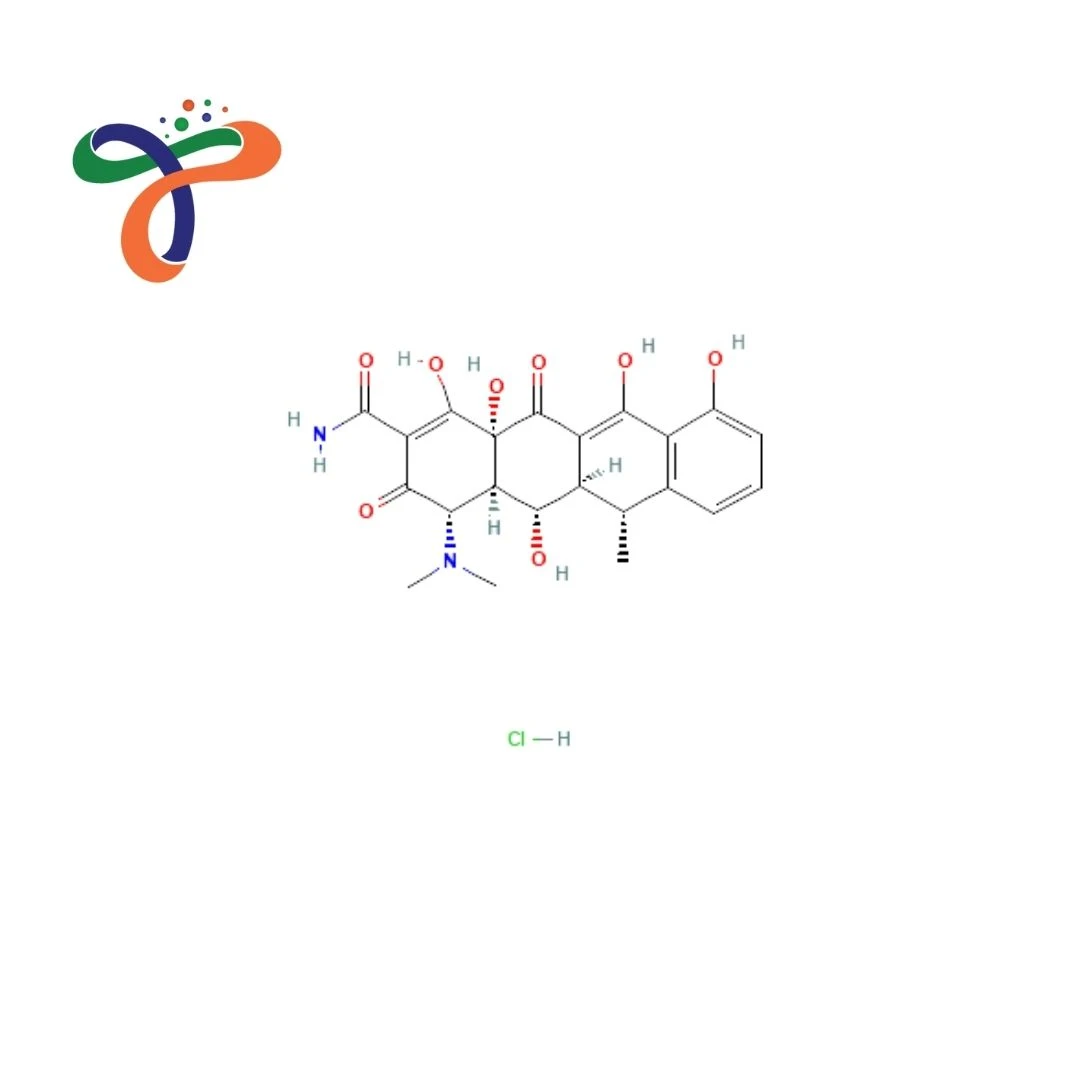
Doxycycline Hydrochloride | 10592-13-9
Cas No :
10592-13-9
Synonyms :
Form :
Powder
Molecular Weight :
480.89 G/Mol
Molecular Formula :
C22H25CLN2O8
Boiling Point :
280 °C
Melting Point :
203 °C
Flash Point :
168 °C
Solubility :
Soluble In Water; Soluble In Methanol; Sparingly Soluble In Ethanol; Insoluble In Diethyl Ether
Description :
Overview of Doxycycline Hydrochloride
Doxycycline Hydrochloride is a pharmaceutical-grade tetracycline-class antibiotic used as an active pharmaceutical ingredient in systemic antibacterial formulations. The hydrochloride salt form improves aqueous compatibility and formulation flexibility for oral dosage forms.
In pharmaceutical manufacturing, Doxycycline Hydrochloride is valued for its broad antimicrobial spectrum, reliable therapeutic performance, and suitability for tablets and capsules. It is produced under GMP-compliant conditions to meet pharmacopeial standards for purity, potency, and safety.
As a pharma chemical, Doxycycline Hydrochloride is widely used in regulated anti-infective drug products.
Applications of Doxycycline Hydrochloride
Broad-Spectrum Antibacterial Formulations
Doxycycline Hydrochloride is used in pharmaceutical products for the management of bacterial infections affecting the respiratory tract, skin, gastrointestinal system, and genitourinary tract. Its wide antimicrobial coverage supports use across multiple therapeutic indications.
Dermatology & Inflammatory Condition Products
Doxycycline Hydrochloride is used in dermatology-focused pharmaceutical formulations where antibacterial and anti-inflammatory effects are required. It supports treatment protocols for acne and chronic inflammatory skin conditions.
Regulated Pharmaceutical Manufacturing Use
Doxycycline Hydrochloride is manufactured as a high-purity pharmaceutical raw material with strict control over moisture content, residual solvents, and impurity profile. It is suitable for large-scale production of regulated oral antibiotic medicines.
In antibiotic formulation portfolios, it is often evaluated alongside Doxycycline Monohydrate, Azithromycin Anhydrous, and Ciprofloxacin Hydrochloride for therapeutic and formulation comparison.
Safety & Handling Guidelines
-
Store in a cool, dry, and controlled environment
-
Protect from light and moisture
-
Keep containers tightly sealed and clearly labelled
-
Avoid inhalation of powder during handling
-
Use appropriate personal protective equipment
-
Handle under controlled pharmaceutical manufacturing conditions
-
Dispose of waste according to regulatory guidelines
Where to Buy Doxycycline Hydrochloride?
Doxycycline Hydrochloride Manufacturer
Doxycycline Hydrochloride is manufactured for pharmaceutical applications where controlled potency, consistent quality, and regulatory compliance are essential.
Doxycycline Hydrochloride Supplier & Distributor
Pharmaceutical-grade Doxycycline Hydrochloride is supplied in suitable packaging for formulation manufacturers. As a pharmaceutical chemicals supplier and distributor, Chemicalbull Pvt. Ltd. supports reliable sourcing with standard documentation.
MSDS for Doxycycline Hydrochloride
The MSDS for Doxycycline Hydrochloride provides information on hazards, handling precautions, storage conditions, exposure controls, and emergency measures. It should be reviewed before pharmaceutical manufacturing use.
Frequently Asked Questions (FAQs)
-
What is Doxycycline Hydrochloride used for?
Doxycycline Hydrochloride is used in pharmaceutical formulations to treat a wide range of bacterial and atypical infections. -
Is Doxycycline Hydrochloride different from Doxycycline Monohydrate?
Yes, both are salt forms of doxycycline with differences in stability, tolerability, and formulation preference. -
What are the side effects of Doxycycline Hydrochloride?
Doxycycline hydrochloride side effects may include gastrointestinal discomfort and photosensitivity depending on dosage and duration.