Benzoic Acid | 65-85-0
Cas No :
65-85-0
Form :
Solid
Molecular Weight :
122.12 G/Mol
Molecular Formula :
C7H6O2
Boiling Point :
249 °C
Melting Point :
122.12 °C
Flash Point :
178 °C
Solubility :
Slightly Soluble In Water; Soluble In Ethanol And Ether
Description :
Overview of Benzoic Acid
Benzoic Acid is a widely used organic compound known for its preservative, antimicrobial, and industrial applications. It is commonly used in food preservation, pharmaceuticals, cosmetics, and chemical manufacturing due to its stability and effectiveness.
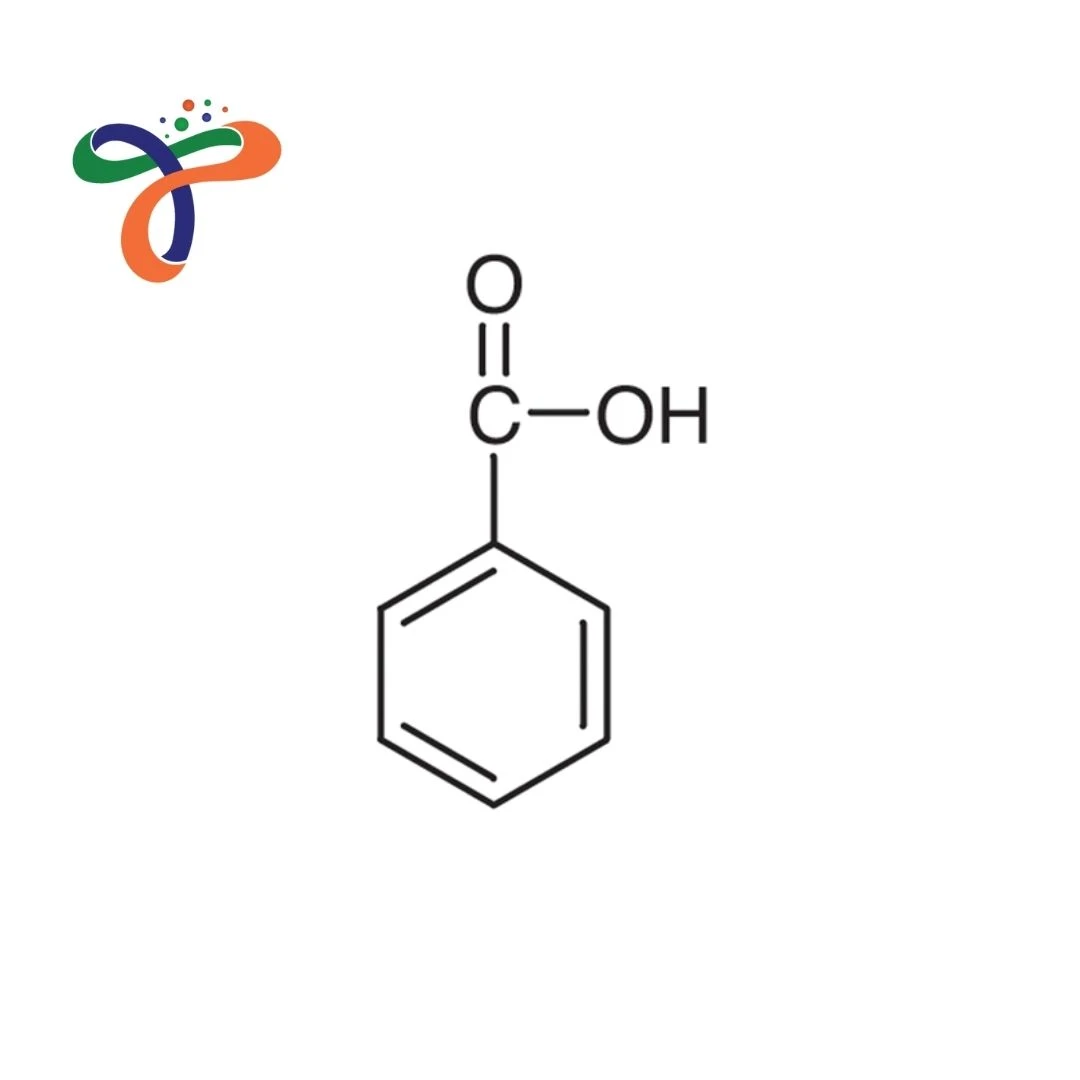
The benzoic acid formula is C₇H₆O₂, with a molecular weight of 122.12 g/mol. It appears as a white crystalline solid with a characteristic odor and has a melting point of approximately 122°C.
As a trusted benzoic acid supplier, manufacturer, and distributor, ChemicalBull provides high-quality bulk material for industrial and global applications.
Applications of Benzoic Acid
Food, Pharmaceutical & Industrial Use
Benzoic acid uses are extensive, especially as a preservative in food and beverages to inhibit microbial growth. It is also used in pharmaceuticals, cosmetics, dyes, and plastic manufacturing.
Understanding how compounds behave chemically, such as which of the following gives benzoic acid on oxidation, is important in industrial synthesis and research applications.
Benzoic acid is often used alongside ingredients like Ascorbic Acid in food and pharmaceutical formulations for enhanced stability and effectiveness.
Chemical Properties & Structure
The benzoic acid structure features a benzene ring bonded to a carboxyl group, making it an aromatic carboxylic acid. The benzoic acid chemical structure explains its acidic behavior and reactivity in organic chemistry.
Due to the electron-withdrawing nature of the carboxyl group, why benzoic acid does not undergo friedel craft reaction is a commonly studied concept — it deactivates the benzene ring toward electrophilic substitution.
Industrial & Formulation Applications
Benzoic Acid is widely used in the manufacture of resins, plasticizers, and alkyd coatings. It also plays a role in personal care and cosmetic formulations due to its antimicrobial properties.
It can be combined with materials like Rapeseed Oil in certain industrial and formulation applications for enhanced performance.
Visit our pharmaceutical chemicals page to explore bulk supply options. Buy, order now, or enquiry now for reliable sourcing.
Safety & Handling Guidelines
-
Store in a cool, dry, well-ventilated place
-
Keep containers tightly sealed to prevent contamination
-
Avoid inhalation of powder; use gloves, goggles, and PPE
-
Handle according to GMP and MSDS guidelines
-
Refer to the MSDS for complete safety, first-aid, and disposal instructions
Where to Buy Benzoic Acid?
Benzoic Acid Manufacturer
As a leading benzoic acid manufacturer in India, Chemicalbull Pvt. Ltd. supplies high-purity material suitable for food, pharmaceutical, and industrial applications.
Benzoic Acid Supplier & Distributor
We are trusted benzoic acid suppliers, distributors, bulk exporters, and global wholesalers, offering consistent quality, documentation, and timely delivery. Buy benzoic acid, purchase now, or contact now for bulk orders.
MSDS for Benzoic Acid
The MSDS provides full information on chemical properties, handling precautions, toxicology, exposure control, and emergency measures for safe use.
Frequently Asked Questions (FAQs)
-
What is benzoic acid used for?
Benzoic acid uses include food preservation, pharmaceuticals, cosmetics, and industrial manufacturing. -
What is the formula and molecular weight of benzoic acid?
The benzoic acid formula is C₇H₆O₂, and the benzoic acid molecular weight is 122.12 g/mol. -
What is the melting point of benzoic acid?
The benzoic acid melting point is approximately 122°C. -
Which compound gives benzoic acid on oxidation?
Compounds like toluene and other alkyl benzenes can give benzoic acid on oxidation under suitable conditions. -
Why does benzoic acid not undergo the Friedel-Crafts reaction?
Because the carboxyl group deactivates the benzene ring, making it less reactive toward electrophilic substitution reactions.