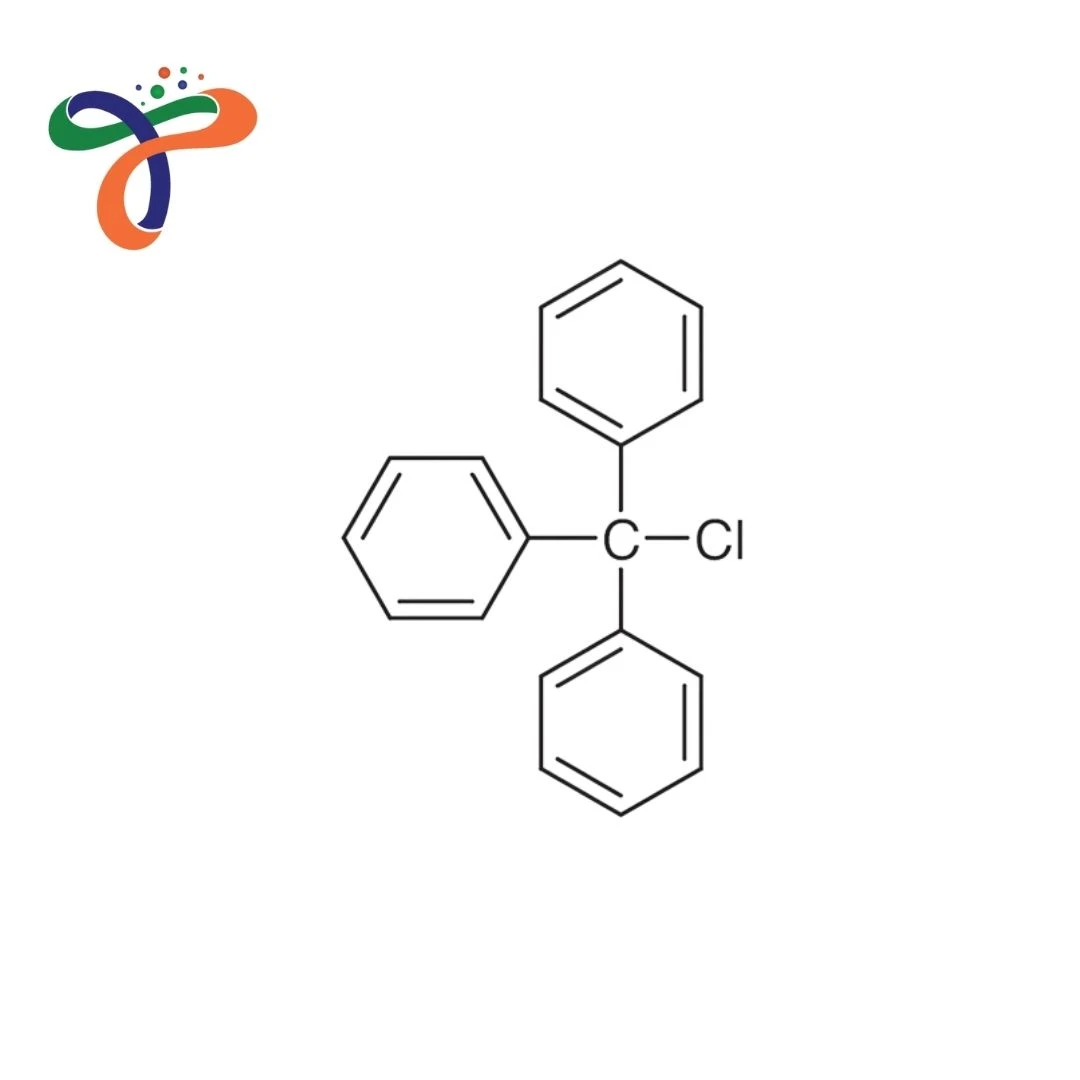
Trityl Chloride | 76-83-5
Cas No :
76-83-5
Molecular Weight :
278.76 G/Mol
Molecular Formula :
C19H15CL
Boiling Point :
280 °C
Melting Point :
66 °C
Flash Point :
60 °C
Solubility :
Insoluble In Water; Soluble In Organic Solvents Such As Dichloromethane, Chloroform, Benzene, Toluene
Description :
Overview of Trityl Chloride
Trityl Chloride (CAS: 2756-39-6) is an organic chloride compound and a highly versatile reagent in the field of organic chemistry. It consists of a trityl group (C₁₇H₁₆) attached to a chlorine atom and is commonly used for the protection of alcohols, phenols, and amines during chemical synthesis. The trityl group (also known as triphenylmethyl group) is a bulky and stable group that provides significant steric hindrance, allowing chemists to selectively protect specific functional groups in organic reactions.
Trityl Chloride is used as a protecting group for alcohols, amines, and carboxylic acids during the synthesis of pharmaceuticals, fine chemicals, and specialty chemicals. It plays an essential role in synthetic organic chemistry by offering a way to block reactive sites in molecules, enabling further reactions to take place without interference.
ChemicalBull offers a range of Trityl Chloride and other protecting group reagents suitable for chemical synthesis in pharmaceutical and laboratory applications.
Applications of Trityl Chloride
Pharmaceutical Synthesis & Drug Development
-
Used as a protecting group in the synthesis of pharmaceutical intermediates and active pharmaceutical ingredients (APIs)
-
Protects alcohols and amines during multistep synthesis of complex drug molecules
-
Facilitates the selective protection of functional groups, allowing for precise reaction control in the pharmaceutical industry
-
Plays a key role in the preparation of targeted drug delivery systems and bioactive compounds
Organic Synthesis & Chemical Reactions
-
Trityl Chloride is extensively used as a protecting group for alcohols, phenols, amines, and carboxylic acids in organic synthesis
-
Facilitates the synthesis of complex organic molecules by temporarily blocking certain reactive sites during chemical reactions
-
Commonly employed in the synthesis of heterocyclic compounds and polymeric materials
-
Used in the preparation of chiral intermediates and fine chemicals for various chemical industries
Laboratory Research & Chemical Engineering
-
Trityl Chloride is a key reagent for scientific research and synthetic chemistry
-
Used in laboratories for the synthesis of novel compounds, material science, and bioorganic chemistry
-
Important in research on organic reactions, reaction mechanisms, and catalysis
Specialty Chemicals & Fine Chemicals
-
Used in the synthesis of specialty chemicals and fine chemicals, including chiral molecules and pharmaceutical precursors
-
Plays a role in polymerization processes and the production of functionalized polymers
-
Utilized in the design and synthesis of new materials and organic products
Safety & Handling Guidelines
-
Wear appropriate protective equipment, such as gloves, goggles, and lab coats to prevent contact with skin and eyes
-
Use in well-ventilated areas to avoid inhalation of vapors or dust
-
Store in tightly sealed containers in a cool, dry place, away from moisture, heat, and oxidizing agents
-
Avoid direct contact with skin and eyes; in case of exposure, wash immediately with water and seek medical attention
-
Dispose of according to local regulations for hazardous chemical waste
Where to Buy Trityl Chloride?
Trityl Chloride Manufacturer
ChemicalBull supplies high-quality Trityl Chloride, suitable for organic synthesis, pharmaceutical manufacturing, and chemical reactions.
Trityl Chloride Supplier & Distributor
-
Available in bulk and laboratory-scale packaging
-
Ideal for chemical protection, pharmaceutical synthesis, and specialty chemical applications
-
COA, MSDS, and technical specifications available
Trityl Chloride MSDS
-
May cause irritation to skin, eyes, and the respiratory system
-
Wear personal protective equipment such as gloves, goggles, and lab coats when handling Trityl Chloride
-
Store away from moisture, oxidizing agents, and sources of heat
-
In case of spill, follow MSDS guidelines for containment and clean-up procedures
-
Refer to MSDS for first aid instructions, fire safety, and spill control
Frequently Asked Questions (FAQs)
-
What is the molecular formula of Trityl Chloride?
Molecular Formula: C₁₇H₁₆Cl
Molecular Weight: 256.77 g/mol -
What is Trityl Chloride used for?
Trityl Chloride is mainly used as a protecting group for amines, alcohols, and carboxylic acids during the synthesis of pharmaceuticals, fine chemicals, and organic compounds. -
Is Trityl Chloride hazardous?
Yes, it may cause irritation to skin, eyes, and the respiratory system. Proper protective equipment should be used when handling. -
How should Trityl Chloride be stored?
Store in airtight containers in a cool, dry place, away from moisture, heat, and oxidizing agents. -
Which industries use Trityl Chloride?
Trityl Chloride is widely used in pharmaceutical synthesis, organic chemistry, research laboratories, chemical engineering, and the production of specialty chemicals.