Lead Tetraacetate | 546-67-8
Cas No :
546-67-8
Form :
Powder
Molecular Weight :
443.38 G/Mol
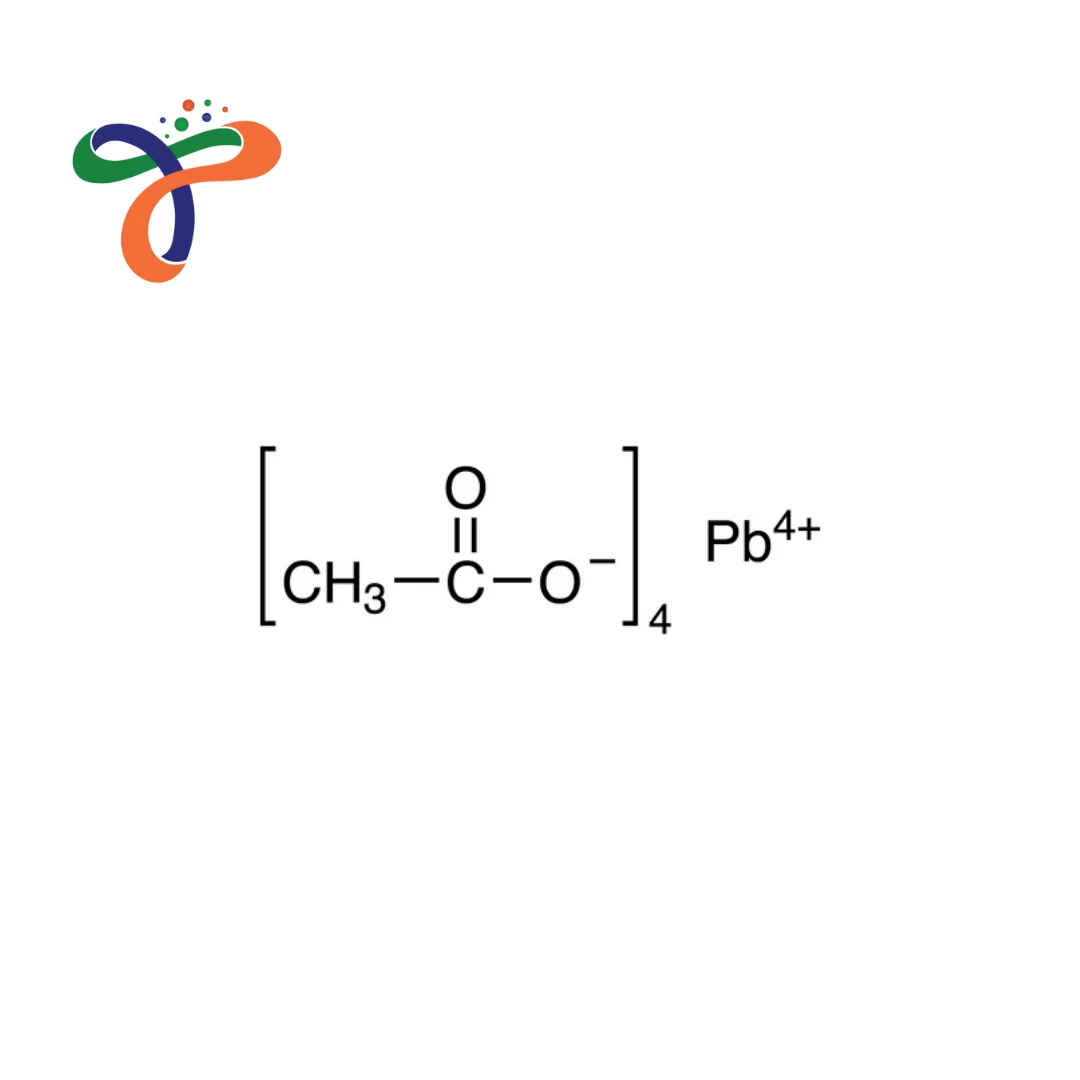
Molecular Formula :
C8H12O8PB
Boiling Point :
350 °C
Melting Point :
110 °C
Flash Point :
120 °C
Solubility :
Soluble In Acetone; Hydrolyzes In Water
Description :
Overview of Lead Tetraacetate
Lead Tetraacetate is an effective antioxidant that is widely employed in organic synthesis and special chemical manufacturing. Organometallic compounds are used in decarboxylation, oxidative cleavage, and functional group transformation reactions. Because of its powerful characteristics, it is mostly used in controlled industrial and laboratory synthetic environments.
The Lead tetraacetate structure consisats of a central lead (Pb⁴⁺) atom coordinated with four acetate (CH₃COO⁻) groups. This tetra-coordinated structure is responsible for its high oxidative reactivity.
Applications of Lead Tetraacetate
Organic Oxidation Reactions
Lead Tetraacetate is widely used as an oxidizing agent in organic chemistry. It is commonly applied in oxidative cleavage of glycols, decarboxylation of carboxylic acids, and selective oxidation of alcohols.
It may be used in reaction systems involving Acetic Acid and other controlled solvent environments to achieve desired transformations.
Pharmaceutical & Fine Chemical Synthesis
In pharmaceutical intermediate production, Lead Tetraacetate is used to modify complex organic molecules through oxidation and rearrangement reactions.
It plays a role in controlled synthesis steps where selective bond cleavage is required.
Lead Tetraacetate Synthesis
Lead tetraacetate synthesis is typically carried out by reacting lead dioxide with acetic acid or acetic anhydride under controlled temperature conditions. The process ensures the formation of the tetravalent lead acetate complex suitable for industrial applications.
Research & Laboratory Applications
The compound is widely used in research labs for research into the mechanisms involved in carbon-carbon bond transformations. Its ability to oxidize makes it an ideal choice for advanced synthetic Chemistry.
Safety & Handling Guidelines
- Store in a cool, dry, and well-ventilated area
- Keep the container tightly sealed
- Avoid inhalation of dust and direct skin contact
- Use protective gloves, goggles, and lab protective clothing
- Handle under a chemical fume hood
- Dispose of waste according to hazardous and environmental regulations (lead-containing compound)
Always consult the official MSDS before handling.
Where to Buy Lead Tetraacetate?
Lead Tetraacetate Manufacturer
Lead Tetraacetate is manufactured under controlled chemical processing conditions to ensure high purity and consistent oxidative performance for industrial and laboratory use.
Lead Tetraacetate Supplier & Distributor
Purchase Lead Tetraacetate from Chemicalbull Pvt. Ltd., a reputable industrial chemical distributor and supplier in India, with reliable bulk supplies and efficient logistical support.
For the latest Lead Tetraacetate price and bulk supply details, contact Chemicalbull Pvt. Ltd. for a competitive industrial quotation.
MSDS for Lead Tetraacetate
The MSDS for Lead Tetraacetate provides detailed information about hazard detection and toxicity data, safe handling methods, storage requirements, exposure control, and emergency response procedures. Industrial users must read this safety information sheet prior to using the product.
Frequently Asked Questions (FAQs)
-
What is Lead Tetraacetate?
Lead Tetraacetate is a strong oxidizing agent used in organic synthesis for oxidative cleavage and functional group transformation reactions. -
What is the Lead tetraacetate formula?
The chemical formula is Pb(C₂H₃O₂)₄. -
What is the Lead tetraacetate structure?
It consists of a central lead atom bonded to four acetate groups. -
What is Lead Tetraacetate used for?
It is used in oxidation reactions, decarboxylation processes, pharmaceutical intermediate synthesis, and laboratory research applications. -
How is Lead tetraacetate synthesis carried out?
It is typically synthesized by reacting lead dioxide with acetic acid or acetic anhydride under controlled conditions.