Thioacetamide
Description :
Overview of Thioacetamide
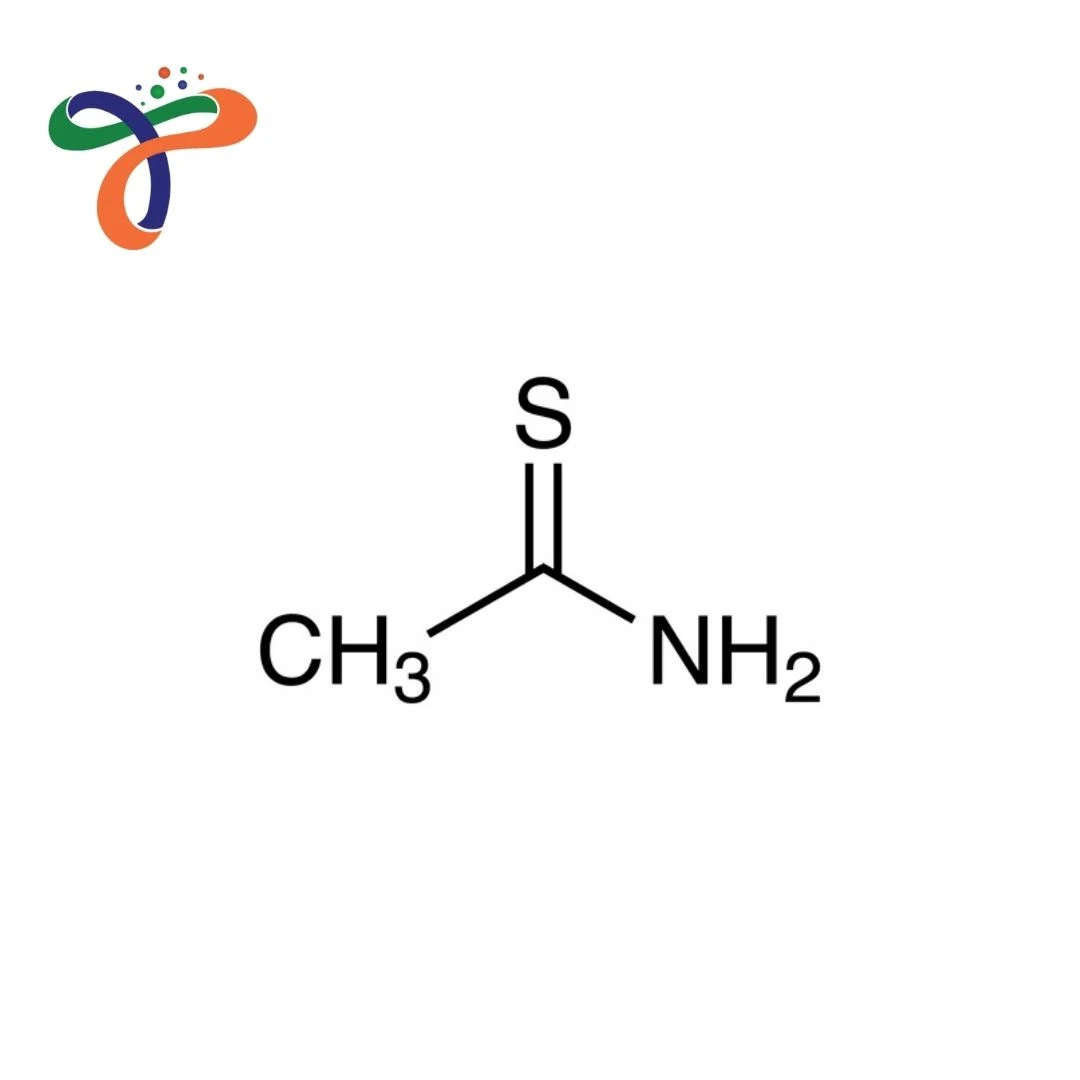
Thioacetamide is an organosulfur compound used as a source of hydrogen sulfide, a reagent in analytical chemistry, and a chemical intermediate in organic synthesis. Known for its use in qualitative analysis for metal precipitation, as a vulcanizing agent, and in the synthesis of heterocyclic compounds, Thioacetamide is widely used in analytical laboratories, industrial chemistry, and pharmaceutical intermediate production.
This compound appears as a white to pale yellow crystalline solid with a characteristic, unpleasant odor. Its thioamide structure provides unique reactivity as a sulfur source in various chemical transformations.
Related products include Thiourea, Sodium Sulfide, and other sulfur-containing reagents used in analytical and synthetic chemistry.
Applications of Thioacetamide
Analytical Chemistry
- Utilized in qualitative analyses to determine the precipitation of metal ions
- It is used to distinguish the metal cations during the systematic study of
- It is a method of obtaining a supply of hydrogen sulfide under controlled conditions
- Utilized in the analytical method for development
- It is used in gravimetric analysis procedures
- It is used in the chemistry classrooms of the educational laboratory
- Utilized in the development of methods for analysis
- It is used for the quality control tests
Industrial Chemical Synthesis
- Used in synthesizing thiazole derivatives
- Applied in preparing heterocyclic compounds
- Used as an intermediate in pharmaceutical synthesis
- Applied in the manufacturing of specialty chemicals
- Used in producing dyes and pigments
- Applied in agrochemical intermediate synthesis
- Used in developing chemical processes
- Applied in fine chemical production
Industrial Applications
- It is used to vulcanize the material for rubber processing
- It is used in leather tanning methods
- used in photographic film development
- It is used in the textile processing industry
- Utilized in the treatment of metals and surface treatment
- Applied in developing industrial formulations
- It is used in polymer chemical applications
- Applied in materials science research
Safety & Handling Guidelines
- TOXIC and CARCINOGENIC—handle with extreme caution
- Avoid inhalation of dust and vapors
- Use only in a well-ventilated fume hood
- Wear appropriate PPE, including gloves, goggles, and a lab coat
- Store in tightly closed containers in a cool, dry place
- Keep away from acids (releases toxic H₂S gas)
- Dispose of according to hazardous waste protocols
- Follow COA and MSDS instructions strictly
Where to Buy Thioacetamide?
Thioacetamide Manufacturer
ChemicalBull supplies high-purity Thioacetamide for authorized analytical and industrial applications with comprehensive safety documentation.
Thioacetamide Supplier & Distributor
- Available in controlled quantities for authorized use
- High-purity analytical and technical grades
- Requires appropriate safety documentation
- COA, MSDS, and comprehensive safety information provided
MSDS
Includes comprehensive toxicity warnings, carcinogenicity information, and strict handling protocols.
Frequently Asked Questions (FAQs)
-
What is Thioacetamide used for?
Used in analytical chemistry for metal precipitation, as a hydrogen sulfide source, in synthesising heterocyclic compounds, and in rubber vulcanisation. -
What is the molecular formula?
Molecular formula: C₂H₅NS or CH₃CSNH₂ with molecular weight 75.13 g/mol. -
Why is Thioacetamide preferred over H₂S gas?
Safer to handle than compressed H₂S gas; provides controlled release of sulfide in solution; easier storage and use in laboratories.