Sodium Polysulfide | 1344-08-07
Cas No :
1344-08-07
Form :
Liquid
Molecular Weight :
174.24 G/Mol
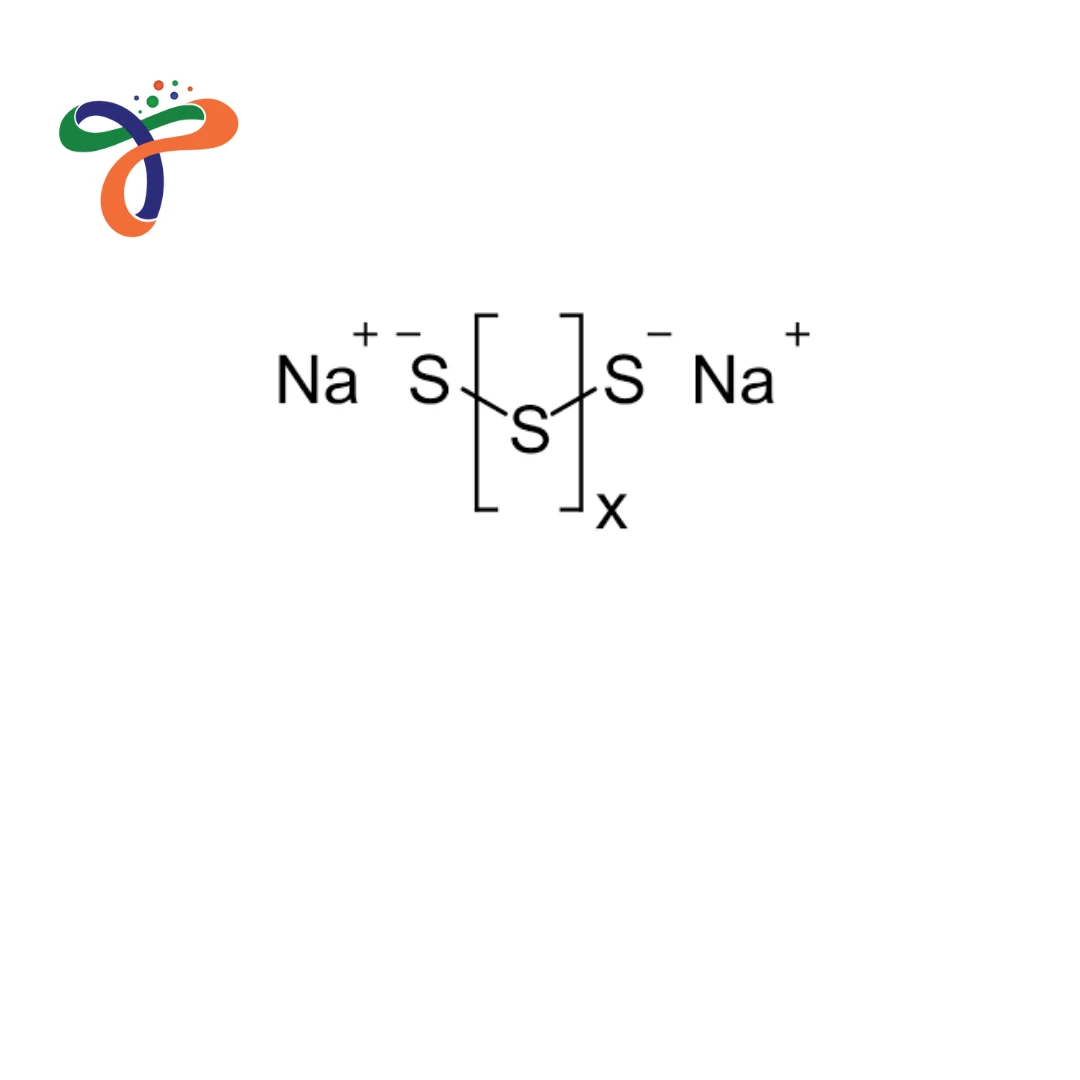
Molecular Formula :
NA2S3
Boiling Point :
380 °C
Melting Point :
140 °C
Flash Point :
135 °C
Solubility :
Water Soluble
Description :
Overview of Sodium Polysulfide
Sodium Polysulfide is an industrial chemical that is sulfur-containing inorganic made up of sodium cations as well as polysulfide anions. It is used extensively in rubber processing, leather manufacturing, the mining process, chemical production, and wastewater treatment. The substance typically appears as a reddish-brown to yellow-colored solid or liquid, depending on the length of the chain and the concentration.
As an industrial chemical, Sodium Polysulfide is valued for its reducing properties, sulfur-donating capability, and reactivity in sulfuration and desulfurization processes. It is supplied in controlled industrial grades for large-scale manufacturing and processing applications.
Applications of Sodium Polysulfide
Leather Processing Industry
Sodium Polysulfide is extensively used in leather tanning and dehairing processes. It assists in removing hair and preparing hides for further treatment in controlled industrial operations.
It may be processed alongside Sodium Sulfide, Lime Powder, and Sodium Hydroxide Prills in structured leather manufacturing systems.
Rubber & Polymer Industry
The compound is employed in special sulfur modification processes in which controlled sulfur incorporation is needed.
It supports industrial rubber compounding applications.
Mining & Ore Flotation
Sodium Polysulfide is applied in flotation processes for metal ore recovery. It enhances selectivity in mineral separation and improves recovery efficiency in mining operations.
It is used in regulated metallurgical environments.
Chemical Synthesis & Waste Treatment
Sodium Polysulfide acts as a sulfurizing agent and reducing agent in specialty chemical manufacturing. It is also used in wastewater treatment for heavy metal precipitation.
It supports industrial environmental management systems.
Safety & Handling Guidelines
- Store in a cool, dry, and well-ventilated area
- Keep containers tightly sealed and protected from moisture
- Avoid inhalation of vapors or fumes
- Prevent contact with skin and eyes
- Use chemical-resistant gloves, goggles, and protective clothing
- Handle under strict industrial safety protocols
- Dispose of waste according to hazardous material regulations
Always consult the SDS before industrial handling.
Where to Buy Sodium Polysulfide?
Sodium Polysulfide Manufacturer
Sodium Polysulfide is manufactured under controlled industrial standards to ensure consistent sulfur content and reliable performance in leather, mining, and chemical industries.
Sodium Polysulfide Supplier & Distributor
The Sodium Polysulfide can be purchased in industrial bulk packaging formats. Purchase Sodium Polysulfide from Chemicalbull Pvt. Ltd., a reputable industrial chemicals distributor and supplier in India, which guarantees reliable logistics and sourcing.
MSDS for Sodium Polysulfide
The MSDS for Sodium Polysulfide provides detailed information on hazard identification, safe handling procedures, storage requirements, exposure controls, and emergency response measures. It must be reviewed prior to use.
Frequently Asked Questions (FAQs)
-
What is Sodium Polysulfide?
Sodium Polysulfide is an inorganic compound consisting of sodium ions and polysulfide (Sₓ²⁻) chains used in industrial sulfur-related processes. -
What is the chemical formula for Sodium Polysulfide?
The general chemical formula is Na₂Sₓ, where “x” represents the number of sulfur atoms in the chain (commonly between 2 and 5). -
What is Sodium Polysulfide used for?
It is used in leather processing, rubber vulcanization, mining flotation, chemical synthesis, and wastewater treatment. -
What is the structure of Sodium Polysulfide?
It consists of sodium cations (Na⁺) associated with linear sulfur chains (Sₓ²⁻), forming ionic polysulfide structures. -
How is Sodium Polysulfide synthesized?
Industrial synthesis typically involves reacting sodium sulfide with elemental sulfur under controlled conditions to form polysulfide chains of varying lengths.