Selenic Acid | 2148941
Cas No :
2148941
Form :
Solid
Molecular Weight :
144.99 G/Mol
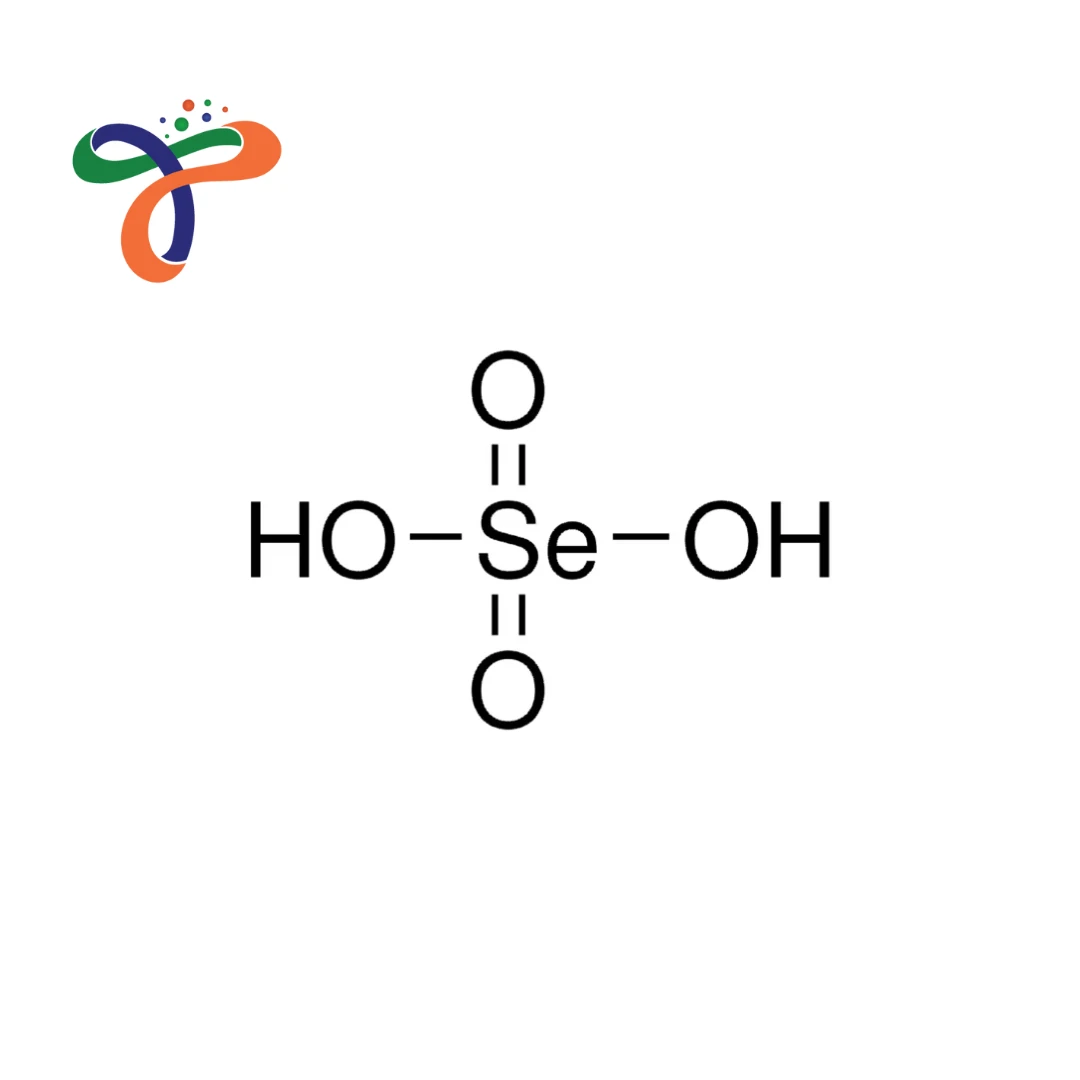
Molecular Formula :
H2O4SE
Boiling Point :
315 °C
Melting Point :
40 °C
Flash Point :
400 °C
Solubility :
Soluble In Water; Miscible
Description :
Overview of Selenic Acid
Selenic Acid (CAS: 7783-00-8) is a highly reactive inorganic selenium compound widely used in oxidizing reactions, analytical chemistry, and specialty chemical synthesis. It appears as a colorless crystalline solid or concentrated solution and is known for its strong oxidative properties, making it a valuable reagent in laboratory and industrial applications.
Selenic Acid is primarily employed in the oxidation of organic compounds, the preparation of selenate salts, and selenium-based chemical research. Its strong oxidizing ability makes it suitable for laboratory experiments, fine chemical manufacturing, and analytical procedures. Related selenium compounds such as Selenious Acid, Sodium Selenate, and Selenium Dioxide are commonly used in specialty chemical synthesis and analytical chemistry.
Applications of Selenic Acid
Analytical Chemistry
-
Used as a strong oxidizing agent for analytical reactions
-
Employed in preparation of selenium-containing standards and reagents
-
Applied in quantitative selenium determination and redox chemistry studies
Laboratory & Research Applications
-
Supports chemical research in selenium chemistry
-
Used in oxidation of organic substrates in laboratory-scale experiments
-
Suitable for academic, industrial, and R&D laboratories
-
Aids in development of novel selenium-based compounds
Specialty Chemical Manufacturing
-
Applied in preparation of selenate salts and selenium-based intermediates
-
Supports fine chemical and reagent production
-
Used in synthesis of high-purity selenium compounds
Industrial Applications
-
Utilized in laboratory and industrial chemical processes
-
Supports manufacture of analytical reagents and specialty chemicals
-
Valuable for selenium research and chemical production
Safety & Handling Guidelines
-
Highly corrosive and toxic; avoid skin, eye, and respiratory exposure
-
Handle in well-ventilated areas or chemical fume hoods
-
Use protective gloves, goggles, and acid-resistant clothing
-
Store in airtight containers, away from reducing agents and organic materials
-
Keep away from heat, moisture, and incompatible substances
-
Follow COA and MSDS instructions strictly
Where to Buy Selenic Acid?
Selenic Acid Manufacturer
ChemicalBull supplies high-purity Selenic Acid suitable for oxidation reactions, analytical chemistry, and specialty chemical synthesis.
Selenic Acid Supplier & Distributor
-
Available in laboratory-scale and industrial bulk packaging
-
Ideal for selenium research, reagent preparation, and fine chemical manufacturing
-
COA, MSDS, and technical specifications provided
Selenic Acid MSDS
-
Causes severe irritation and burns to skin, eyes, and respiratory tract
-
Strong oxidizer—may react violently with organic materials
-
Handle with proper PPE and ventilation
-
Store in cool, dry, well-ventilated areas away from incompatible chemicals
-
Refer to full MSDS for emergency, spill, and disposal procedures
Frequently Asked Questions (FAQs)
-
What is the molecular formula of Selenic Acid?
Molecular Formula: H₂SeO₄
Molecular Weight: 128.97 g/mol -
What is Selenic Acid used for?
Used primarily as a strong oxidizing agent, in selenium-based research, and for preparation of selenate salts. -
Is Selenic Acid hazardous?
Yes. It is highly corrosive and toxic. Proper PPE and safety precautions are mandatory.