Boron Trichloride | 10294-34-5
Cas No :
10294-34-5
Form :
Liquid
Molecular Weight :
117.16 G/Mol
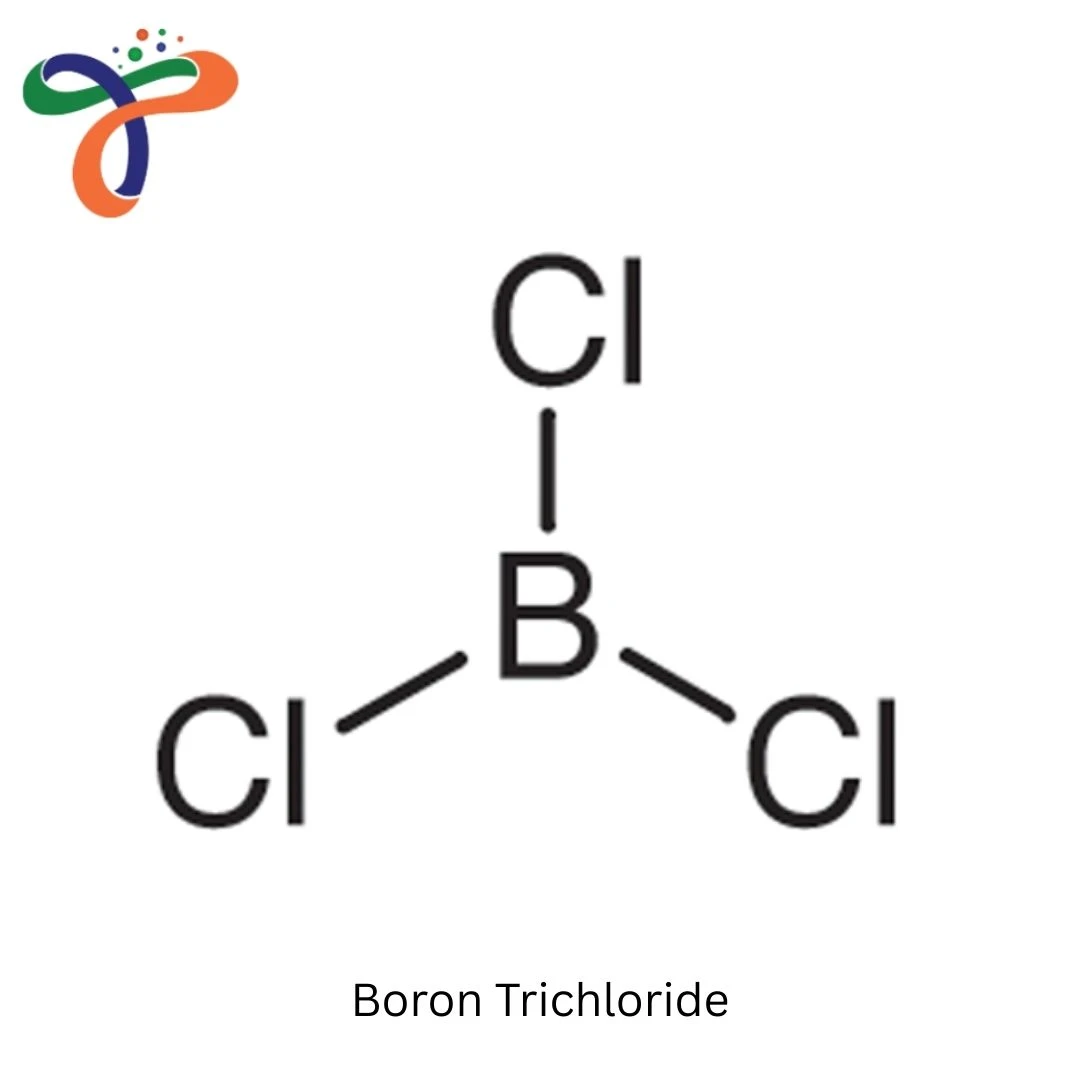
Molecular Formula :
BCL3
Boiling Point :
-85.1 °C
Melting Point :
-133.0 °C
Flash Point :
-120.0 °C
Solubility :
Reacts With Water; Hydrolyzes To Hcl And Boric Acid; Soluble In Nonpolar Solvents
Description :
Overview of Boron Trichloride
Boron Trichloride (BCl3) can be described as an extremely reactive industrial inorganic compound that is widely used as a catalyst for Lewis acids and a chemical intermediate. It is usually used in compressed gas form or compressed gas that is liquefied under pressure to be used in industrial applications.
Being a powerful electron-pair acceptor Boron Trichloride is a key component in organic chemical synthesis, petrochemical processing semiconductor manufacturing, and special chemical production. Because of its high reactivity and sensitivity to moisture it is required to be handled under strict control industrial conditions.
Boron Trichloride is considered an essential reagent in advanced chemical manufacturing processes where catalytic activation and halogenation reactions are required.
Applications of Boron Trichloride
Catalyst in Organic & Petrochemical Industries
Boron Trichloride is widely used as a Lewis acid catalyst in alkylation, polymerization, and dealkylation reactions. It is applied in petrochemical refining and hydrocarbon processing.
In catalytic systems, it may be used alongside Aluminium Chloride Anhydrous, Sulfuric Acid, and Phosphorus Oxychloride to support high-efficiency industrial reactions.
Semiconductor & Electronics Manufacturing
BCl₃ is extensively used in plasma etching processes for semiconductor fabrication. It helps remove metal oxides and is critical in integrated circuit production.
It may be processed in controlled systems with specialty gases and chemicals such as Hydrochloric Acid, Chlorine, and Silicon Tetrachloride, depending on the application.
Pharmaceutical & Fine Chemical Intermediate
Boron Trichloride is used in organic synthesis for demethylation and other functional group transformations. It plays a role in producing active pharmaceutical intermediates and specialty fine chemicals.
Polymer & Specialty Chemical Manufacturing
Boron Trichloride is utilized in organic synthesis for demethylation as well as different functional group transformations. It is involved in the production of active pharmaceutical intermediates as well as specialty fine chemicals.
Safety & Handling Guidelines
- Keep in pressure-rated cylinders with a valid approval
- Stay clear of the sources of water and moisture.
- Use in controlled, well-ventilated industrial settings
- Avoid inhalation of gas
- Keep away from contact with eyes and skin
- Wear appropriate respiratory protection, gloves, and a face shield
- Handle only under the supervision of a trained professional.
- Follow the regulations for transport and disposal
Always review the MSDS before industrial handling.
Where to Buy Boron Trichloride?
Boron Trichloride Manufacturer
Boron Trichloride is manufactured for industrial-grade applications requiring high purity and consistent reactivity, particularly in semiconductor and catalytic industries.
Boron Trichloride Supplier & Distributor
Boron Trichloride is supplied in compressed gas cylinders suitable for industrial use. Buy Boron Trichloride from Chemicalbull Pvt. Ltd., a trusted industrial chemicals supplier and distributor in India, offering reliable sourcing and compliant logistics.
MSDS for Boron Trichloride
The MSDS for Boron Trichloride provides comprehensive information regarding hazard identification and safe handling practices, emergency response measures, storage requirements, and exposure control measures. It should be reviewed prior to using.
Frequently Asked Questions (FAQs)
-
What is Boron Trichloride used for?
Boron Trichloride is used as a Lewis acid catalyst in petrochemical processing, semiconductor etching, organic synthesis, polymer manufacturing, and specialty chemical production. -
Is BCl₃ stronger than BF₃?
Yes, Boron Trichloride (BCl₃) is generally considered a stronger Lewis acid than Boron Trifluoride (BF₃) because it experiences less effective back-donation from chlorine atoms compared to fluorine in BF₃. -
What is the reaction of Boron Trichloride with water?
Boron Trichloride reacts vigorously with water, producing hydrochloric acid (HCl) and boric acid derivatives. This reaction releases heat and corrosive fumes, which is why moisture-free handling is essential.