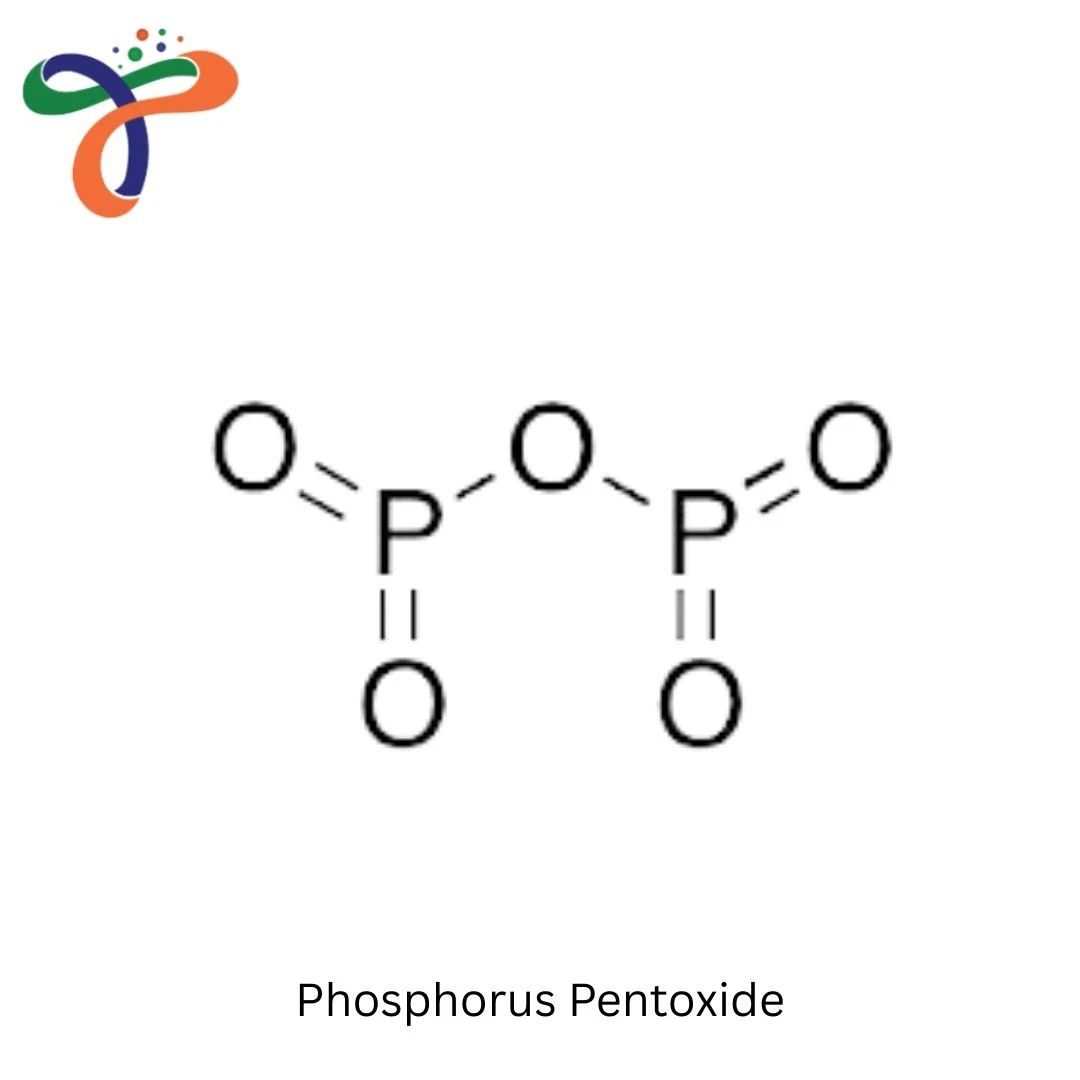
Phosphorus Pentoxide | 1314-56-3
Cas No :
1314-56-3
Form :
Powder
Molecular Weight :
141.94 G/Mol
Molecular Formula :
O5P2
Boiling Point :
360 °C
Melting Point :
340 °C
Flash Point :
360 °C
Solubility :
Reacts Vigorously With Water To Form Phosphoric Acid
Description :
Phosphorus Pentoxide
Phosphorus Pentoxide, also known as Phosphoric Anhydride, is one of the most powerful dehydrating and condensing agents available in industrial chemistry. As a leading raw chemical supplier, Triveni Chemicals provides high-purity Phosphorus Pentoxide in a stable, white crystalline powder form, specifically manufactured for high-performance chemical synthesis and laboratory applications.
We support a diverse range of industries across India and the global market, ensuring a reliable supply of this highly reactive industrial reagent from our strategic base in the Vapi/Valsad industrial belt. We are a trusted Phosphorus Pentoxide manufacturer, providing high-purity P_2O_5 powder for diverse chemical applications. Known as a superior dehydrating agent, our product is essential for moisture removal in organic synthesis and industrial processes.
Key Technical Specifications:
Triveni Chemicals ensures our Phosphorus Pentoxide meets the rigorous standards required for moisture-sensitive chemical processes:
- Chemical Name: Phosphorus Pentoxide (Phosphoric Anhydride)
- Chemical Formula: P4O10
- Appearance: White, deliquescent crystalline powder
- Density: 2.39 g/cm³
- Melting Point: 340°C (under pressure)
- Solubility: Reacts violently with water to form phosphoric acid
- CAS Number: 1314-56-3
Caution: Phosphorus Pentoxide is highly corrosive and reacts exothermically with moisture. Please refer to the technical safety data provided by Triveni Chemicals.
Industrial & Commercial Applications
Phosphorus Pentoxide is prized for its extreme affinity for water, making it indispensable in several specialized sectors:
- Dehydrating Agent: Utilized in laboratories and industrial plants to remove trace amounts of water from solvents and gases, achieving levels of dryness that other desiccants cannot reach.
- Chemical Synthesis: Acts as a vital reagent in the preparation of esters, acid anhydrides, and phosphorus-based organic compounds.
- Phosphate Ester Production: Used as a primary raw material in the manufacturing of surfactants, flame retardants, and plasticizers.
- Specialty Glass Manufacturing: Incorporated into the production of optical glass and heat-resistant glass to improve durability and refractive properties.
- Asphalt Modification: Employed in the air-blowing process of bitumen to improve the viscosity and temperature susceptibility of asphalt used in road construction.
Explore our industrial applications page to see how our high-grade reagents can optimize your specific chemical workflows.
Why Partner with Triveni Chemicals?
At Triveni Chemicals, we bridge the gap between technical grade requirements and logistical efficiency:
- High Reactivity Standards: We ensure our Phosphorus Pentoxide is packed in moisture-proof containers to maintain its potency from our facility to your production line.
- Global Export Reach: Operating from the heart of Gujarat's industrial hub, we manage secure logistics for domestic and international hazardous material transport.
- Technical Quality Control: Every batch is tested for purity and particle consistency, ensuring predictable results in your sensitive synthesis reactions.
Seeking a high-performance dehydrating agent?
Request a quote today to discuss your bulk volume requirements or explore the full product catalog at Triveni Chemicals.
Frequently Asked Questions (FAQ)
-
What is Phosphorus Pentoxide used for?
It is primarily used as a powerful dehydrating agent, a chemical intermediate for phosphoric acid, and a high-efficiency desiccant. -
Is Phosphorus Pentoxide hazardous?
Yes, it is a highly corrosive and reactive substance that reacts violently with water to form phosphoric acid. Proper PPE and dry storage are mandatory. -
What is the CAS number for P2O5?
The standard CAS number for Phosphorus Pentoxide is 1314-56-3.