Nitric Acid | 7697-37-2
Cas No :
7697-37-2
Form :
Liquid
Molecular Weight :
63.01
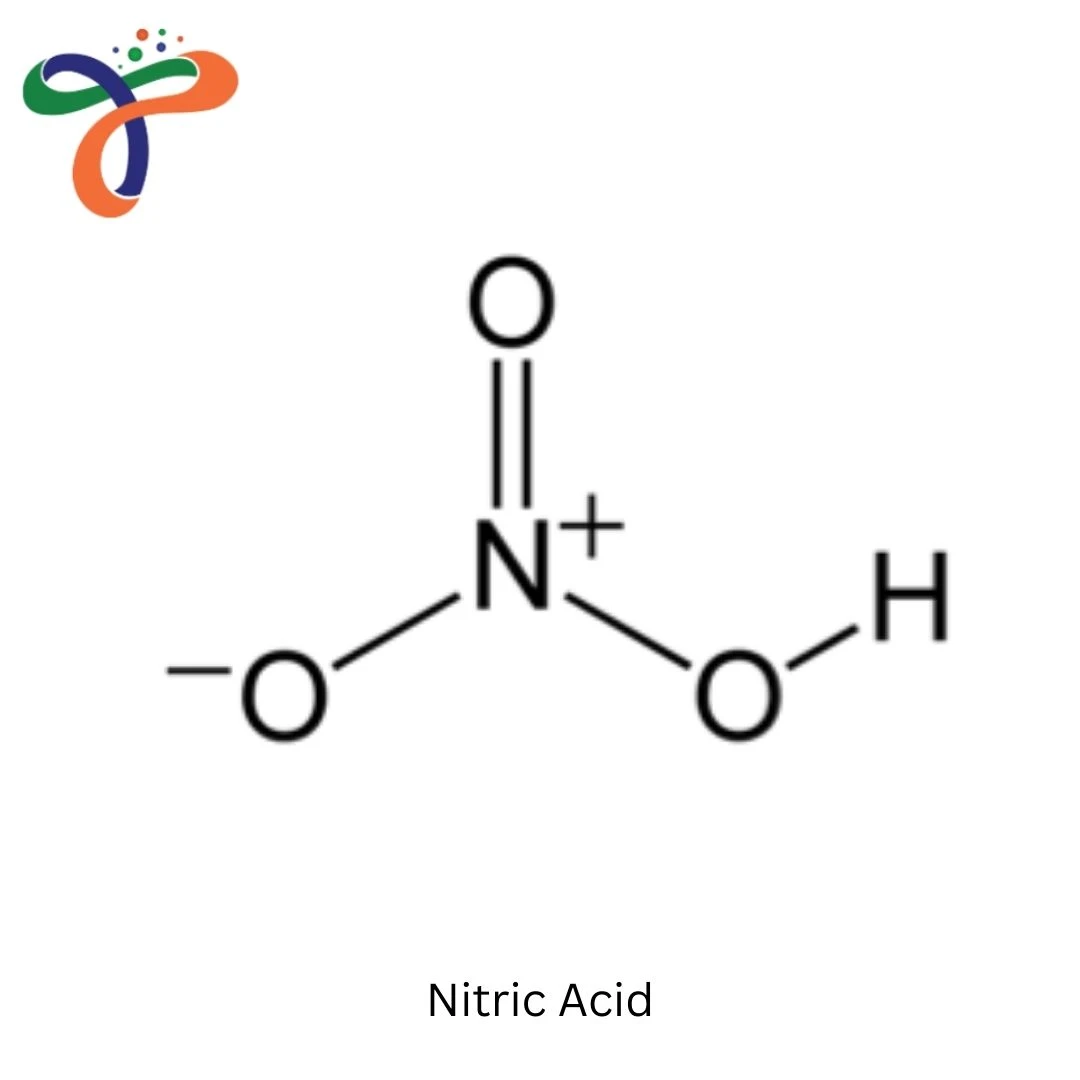
Molecular Formula :
HNO3
Boiling Point :
121 °C
Description :
Overview of Nitric Acid
Nitric Acid (HNO₃) is a strong mineral acid widely used in agrochemical applications such as fertilizer production, chemical manufacturing, and metal processing. It is a highly reactive and corrosive chemical known for its role as a powerful oxidizing agent.
Nitric Acid is known for its sharp, acrid smell and is usually produced in concentrated forms. It plays a key role in various chemical processes, and its wide range of applications makes it a fundamental chemical in the global industrial sector.
Whether you are seeking a Nitric Acid manufacturer for high-volume ammonium nitrate production or for specialized laboratory reagents, our material ensures a consistent performance profile. To explore how our grades can optimize your specific chemical process, you can connect with our technical team directly to discuss bulk supply and current market pricing.
Applications of Nitric Acid
Nitric Acid is commonly used in fertilizer production, explosives manufacturing, metal treatment, and chemical synthesis. It plays a crucial role in producing ammonium nitrate, cleaning metals, and industrial chemical reactions.
-
Fertilizer Industry: The primary use for Nitric Acid is the production of Ammonium Nitrate, Urea, and Calcium Ammonium Nitrate, which are indispensable for boosting global agricultural productivity.
-
Explosives Manufacturing: A critical component in the synthesis of industrial explosives like TNT (Trinitrotoluene) and nitroglycerin through the nitration of organic compounds.
-
Metal Treatment & Finishing: Utilized for pickling, descaling, and cleaning metal surfaces. It effectively removes impurities, preparing stainless steel and other alloys for further processing.
-
Chemical Synthesis: Acts as a vital reagent in the production of adipic acid (for nylon), nitrobenzene, and various organic nitrates used in industrial chemistry.
-
Laboratory Applications: Widely used in research for qualitative analysis, metal digestion, and as a strong oxidizing agent in chemical reactions.
-
Electronics Industry: Employed in the etching of printed circuit boards (PCBs) and cleaning of semiconductor wafers.
Safety & Handling Guidelines
Due to its highly corrosive nature and oxidizing power, professional handling is mandatory:
- Protective Gear: Always wear acid-resistant gloves, chemical-proof aprons, and full-face shields to prevent severe skin and eye burns.
- Inhalation Prevention: Avoid breathing fumes; operate only in areas with high-efficiency fume hoods or localized exhaust systems.
- Chemical Compatibility: Keep strictly away from organic materials, fuels, and metallic powders, as contact can cause spontaneous combustion or explosions.
- Hygiene: Maintain strict personal safety protocols; emergency showers and eyewash stations must be accessible in handling zones.
- Spill Management: Neutralize small spills with soda ash or lime only after ensuring adequate ventilation to manage heat and gas release.
Where to Buy Nitric Acid?
Nitric Acid Manufacturer
We are a leading Nitric Acid manufacturer, providing high-quality nitric acid for various industrial applications, including fertilizer production, explosives manufacturing, and metal treatment.
Nitric Acid Supplier & Distributor
We are a trusted Nitric Acid supplier in India, providing high-quality acid for industrial applications. Businesses looking to buy Nitric Acid in bulk can contact us for competitive pricing and reliable supply. The Nitric Acid price depends on concentration and order quantity. We offer bulk supply with consistent quality, secure packaging, and fast delivery for industrial use.
MSDS for Nitric Acid
The MSDS for Nitric Acid provides crucial safety information regarding handling, storage, and emergency procedures. Always refer to the MSDS to ensure safe handling of the material.
Related Products
-
Sulfuric Acid – Frequently used alongside Nitric Acid in nitration mixtures for explosives and dyes.
-
Sodium Persulfate – A related oxidizing agent used in electronics etching and polymer synthesis.
-
Phosphoric Acid – Utilized in metal treatment and fertilizer production.
-
Sodium Gluconate Manufacturer – Often used as a chelating agent in alkaline cleaning formulations.
Frequently Asked Questions (FAQs)
-
What is the formula of Nitric Acid?
The chemical formula of Nitric Acid is HNO₃, which represents the compound consisting of one hydrogen atom, one nitrogen atom, and three oxygen atoms. -
What is the molecular weight of Nitric Acid?
The molecular weight of Nitric Acid is 63.01 g/mol. -
What happens when eggshell is added to Nitric Acid?
When eggshell (calcium carbonate) is added to Nitric Acid, it reacts to produce calcium nitrate, carbon dioxide, and water. -
Does Nitric Acid turn red litmus blue?
True or False: Nitric Acid is an acid, so it turns red litmus paper blue, making the statement true. -
How is Nitric Acid formed?
Nitric Acid is primarily produced via the Ostwald process, where ammonia (NH₃) is oxidized to form nitric oxide (NO), which is then further oxidized to form nitrogen dioxide (NO₂). The nitrogen dioxide is then absorbed in water to produce Nitric Acid.