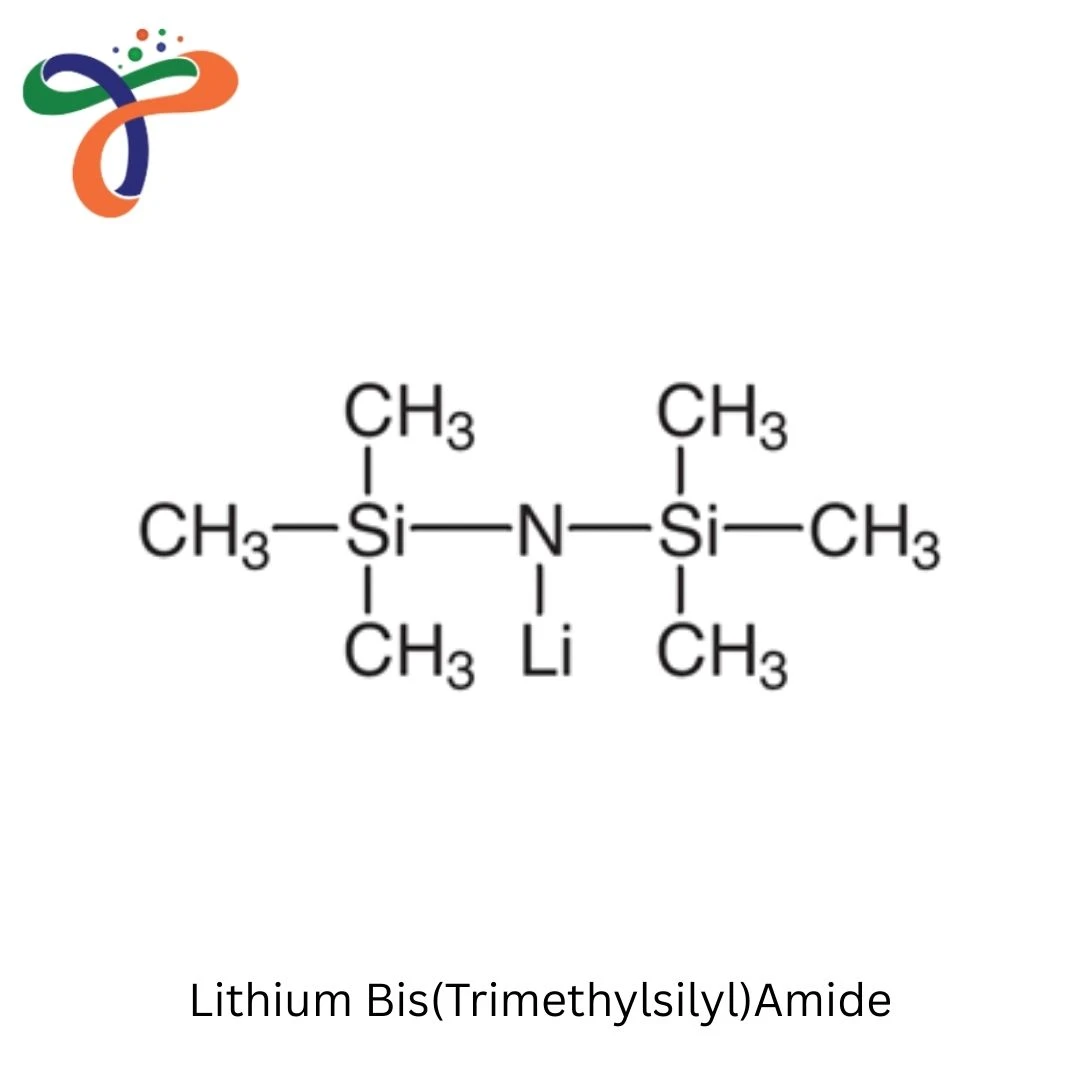
Lithium Bis(Trimethylsilyl)Amide | 4039-32-1
Cas No :
4039-32-1
Form :
Powder
Molecular Weight :
167.33 G/Mol
Molecular Formula :
C6H19LINSI2
Boiling Point :
200 °C
Melting Point :
160 °C
Flash Point :
200 °C
Solubility :
Soluble In Thf And Diethyl Ether; Insoluble In Water.
Description :
Overview of Lithium Bis(Trimethylsilyl)Amide
Lithium Bis(Trimethylsilyl)Amide (LiHMDS, LiN(TMS)₂) is a strong, non-nucleophilic base used extensively in organic synthesis, pharmaceutical manufacturing, and polymer chemistry. Known for its excellent basicity without nucleophilicity, steric bulk preventing side reactions, and ability to generate enolates and other reactive intermediates, LiHMDS is widely used in pharmaceutical synthesis, materials chemistry, and fine chemical production.
This compound appears as a white to off-white powder or is supplied as a solution in various solvents (THF, hexanes). Its large trimethylsilyl groups provide steric hindrance, making it an excellent non-nucleophilic strong base.
Related products include Sodium Bis(trimethylsilyl)amide (NaHMDS), Potassium Bis(trimethylsilyl)amide (KHMDS), Methylmagnesium Bromide, and other strong bases used in organic synthesis. Works complementarily with Boron Tribromide and Cyclohexene Oxide in various synthetic transformations.
Applications of Lithium Bis(Trimethylsilyl)Amide
Pharmaceutical Synthesis
- Used in generating enolates for aldol reactions
- Applied in pharmaceutical intermediate synthesis
- Used in stereoselective synthesis
- Applied in preparing drug candidates
- Used in manufacturing active pharmaceutical ingredients
Industrial Organic Synthesis
- Used in deprotonation reactions
- Applied in anionic polymerization initiation
- Used in preparing organometallic compounds
- Applied in synthesizing specialty chemicals
Advanced Chemical Transformations
- Used in C-C bond-forming reactions
- Applied in Claisen condensations
- Used in Michael's additions
- Applied in synthesizing heterocyclic compounds
Safety & Handling Guidelines
- HIGHLY REACTIVE and PYROPHORIC—handle with extreme care
- Reacts violently with water, oxygen, and protic solvents
- Use only under strictly anhydrous conditions and an inert atmosphere
- Handle in the glove box or using Schlenk techniques
- Wear full PPE, including chemical-resistant gloves, face shield
- Store in tightly sealed containers under argon or nitrogen
Where to Buy Lithium Bis(Trimethylsilyl)Amide?
Lithium Bis(Trimethylsilyl)Amide Manufacturer
ChemicalBull supplies high-purity Lithium Bis(Trimethylsilyl)Amide for pharmaceutical and research applications.
Lithium Bis(Trimethylsilyl)Amide Supplier & Distributor
- Available as powder or solution in THF, hexanes, toluene
- Multiple concentration options (1M, 1.5M in various solvents)
- High-purity grades for pharmaceutical synthesis
- Packaged under an inert atmosphere with appropriate safety features
- COA, MSDS, and comprehensive safety documentation provided
MSDS
Includes comprehensive pyrophoric material handling instructions, reactivity warnings, and emergency procedures.
Frequently Asked Questions (FAQs)
-
What is Lithium Bis(Trimethylsilyl)Amide used for?
Used as a strong non-nucleophilic base for deprotonation, generating enolates, in pharmaceutical synthesis, anionic polymerization, and various organic transformations
. -
What is the molecular formula?
Molecular formula: C₆H₁₈LiNSi₂ or LiN(Si(CH₃)₃)₂ with molecular weight 167.32 g/mol. -
How does LiHMDS differ from other strong bases?
Bulky trimethylsilyl groups provide steric hindrance, preventing nucleophilic attack; excellent for generating enolates without competing nucleophilic reactions.