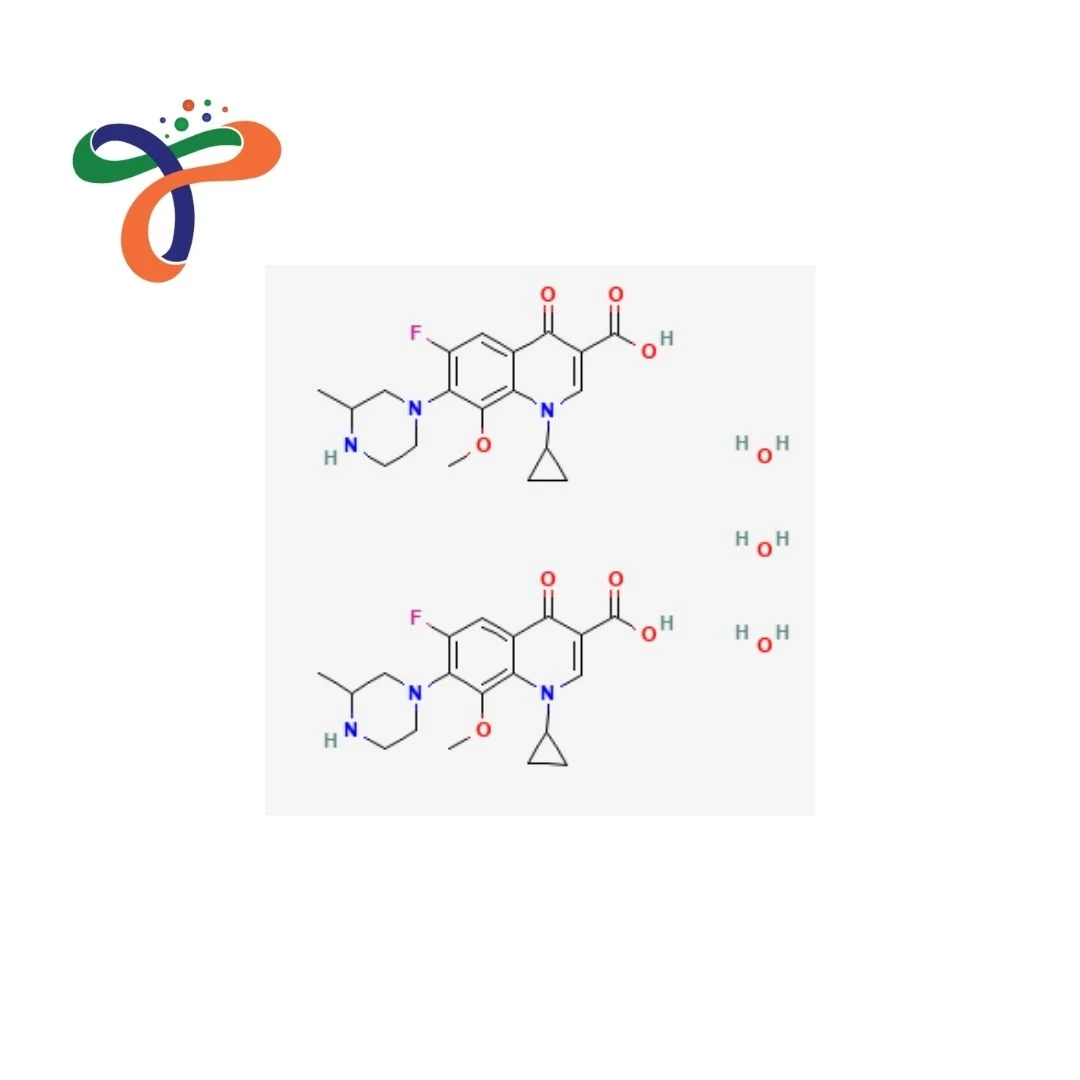
Gatifloxacin Sesquihydrate | 180200-66-2
Cas No :
180200-66-2
Form :
Powder
Molecular Weight :
419.50 G/Mol
Molecular Formula :
C38H50F2N6O11
Boiling Point :
360 °C
Melting Point :
230 °C
Flash Point :
160 °C
Solubility :
Soluble In Water And Methanol; Soluble In Dmso
Description :
Overview of Gatifloxacin Sesquihydrate
Gatifloxacin Sesquihydrate is a pharmaceutical-grade fluoroquinolone antibacterial that is supplied in the form of an active pharmaceutical ingredient (API). It is the squihydrate variant of gatifloxacin. It was designed to ensure stability controlled and to be suitable for formulations in pharmaceuticals.
In the manufacturing of pharmaceuticals, Gatifloxacin Sesquihydrate is valued due to its solid-state properties, consistent antibacterial activity, and compatibility with both oral and ophthalmic dosage forms. It is manufactured under controlled conditions that meet the pharmacopeial requirements regarding purity, hydrolysis status, and chemical identification.
As a pharma chemical, Gatifloxacin Sesquihydrate is handled exclusively within licensed pharmaceutical manufacturing and formulation environments.
Applications of Gatifloxacin Sesquihydrate
Pharmaceutical API Use
Gatifloxacin Sesquihydrate is utilized as a pharmaceutical active ingredient in the production of antibacterial formulas, notably oral dosage forms of solids and Ophthalmic preparations. The fluoroquinolone action of this ingredient aids in the development of new products to treat bacteria that are infected under controlled pharmaceutical usage.
In antibacterial API portfolios, Gatifloxacin Sesquihydrate is commonly positioned alongside Ciprofloxacin Hydrochloride, Levofloxacin, and Moxifloxacin Hydrochloride for fluoroquinolone-based formulation development.
Finished Dosage Form Manufacturing
Gatifloxacin Sesquihydrate is used in the production of tablets and eye-drop formulations where controlled dissolution, stability, and dose uniformity are critical. The sesquihydrate form contributes to reproducible manufacturing performance and shelf stability.
Manufacturing is carried out under GMP-compliant conditions due to the potency and regulatory status of the compound.
Pharmaceutical Research & Reference Use
Gatifloxacin Sesquihydrate is also used as a reference chemical in research in the field of pharmaceuticals, as well as analytical method validation and testing for quality control. It is used for solubility profiling and stability studies, as well as comparisons of antibacterial testing.
In research environments, it may be referenced alongside Ofloxacin and Norfloxacin for fluoroquinolone comparison studies.
Safety & Handling Guidelines
- Store in a cool, dry, and controlled environment
- Protect from light and moisture
- Keep containers tightly sealed and properly labelled
- Avoid inhalation or direct contact with API powder
- Wear appropriate personal protective equipment
- Only handle in areas of pharmaceutical manufacturing
- Waste disposal is governed by guidelines for pharmaceutical regulation
Where to Buy Gatifloxacin Sesquihydrate?
Gatifloxacin Sesquihydrate Manufacturer
Gatifloxacin Sesquihydrate is manufactured for pharmaceutical use under strict GMP conditions to ensure consistent quality, hydration control, and regulatory compliance.
Gatifloxacin Sesquihydrate Supplier & Distributor
Pharmaceutical-grade Gatifloxacin Sesquihydrate API is supplied in compliant packaging suitable for licensed pharmaceutical manufacturers. As a pharmaceutical chemicals supplier and distributor, Chemicalbull Pvt. Ltd. supports reliable sourcing with COA, MSDS, and regulatory documentation.
MSDS for Gatifloxacin Sesquihydrate
The MSDS for Gatifloxacin Sesquihydrate contains detailed information about chemicals, safety handling procedures, specifications for storage and exposure control, and emergency response procedures. It should be reviewed prior to making or using it in the laboratory.
Frequently Asked Questions (FAQs)
-
What is Gatifloxacin Sesquihydrate?
Gatifloxacin Sesquihydrate is a fluoroquinolone antibiotic supplied as a pharmaceutical-grade active pharmaceutical ingredient in its sesquihydrate solid form. -
What is the solubility of Gatifloxacin Sesquihydrate?
Gatifloxacin Sesquihydrate is sparingly soluble in water and shows improved solubility in acidic aqueous media. Its solubility profile is an important consideration during formulation development and dissolution optimisation. -
What is the formula of Gatifloxacin Sesquihydrate?
The formula of Gatifloxacin Sesquihydrate is C38H50F2N6O11.