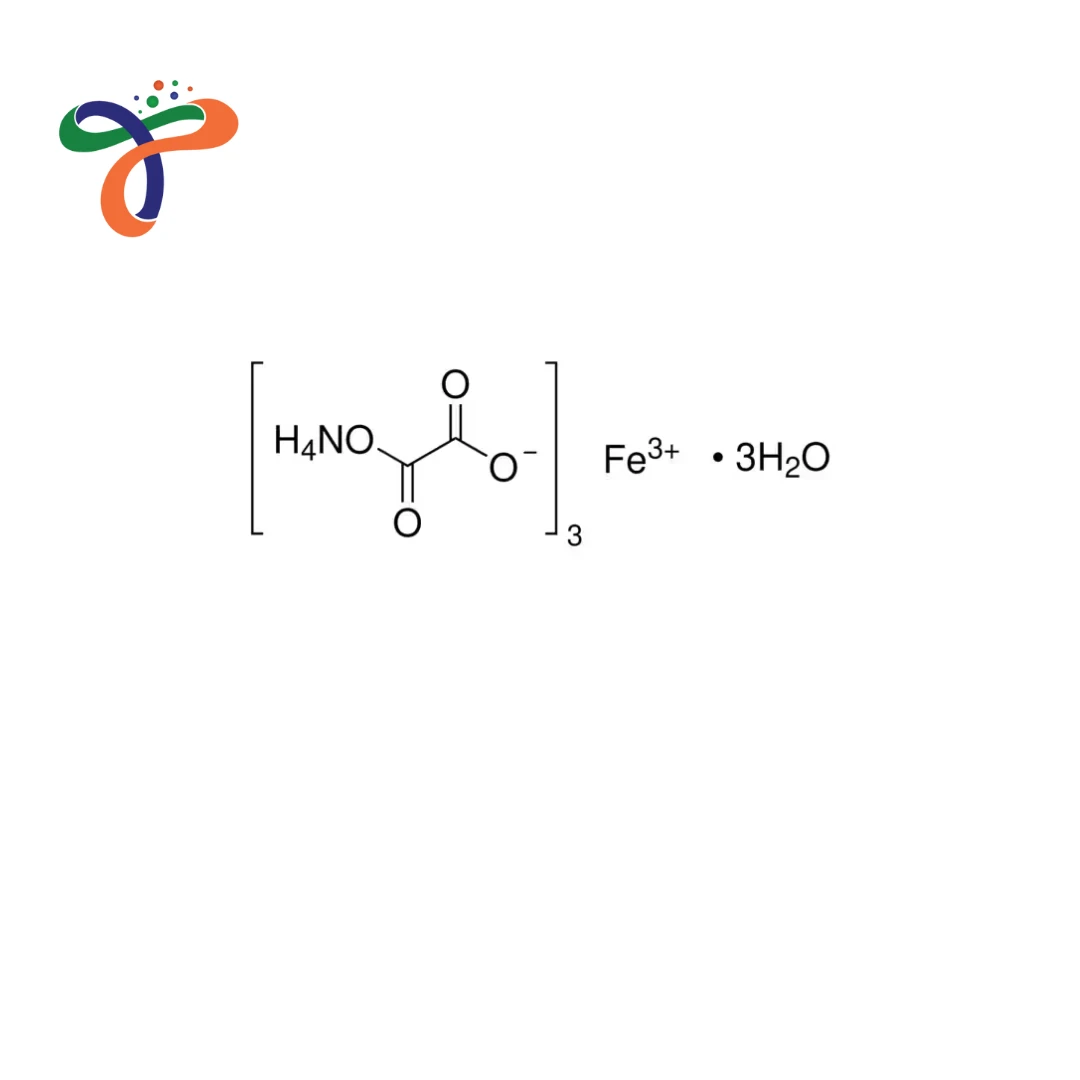
Ferric Ammonium Oxalate | 14221-47-7
Cas No :
14221-47-7
Form :
Solid
Molecular Weight :
249.92 G/Mol
Molecular Formula :
C6FEO12.3H4N
Boiling Point :
600 °C
Melting Point :
110 °C
Flash Point :
250 °C
Solubility :
Soluble In Water; Sparingly Soluble In Ethanol
Description :
Overview of Ferric Ammonium Oxalate
Ferric Ammonium Oxalate is an iron coordination compound used in photography, blueprint processes, and analytical chemistry. Known for its light-sensitive properties, use in photographic printing, and applications as an analytical reagent, Ferric Ammonium Oxalate is widely used in photographic processes, developing blueprints, analytical chemistry, and specialty applications.
This compound appears as a green crystalline solid. Its iron(III) oxalate complex structure provides useful photosensitive and analytical properties.
Related products include Ferric Oxalate, Ammonium Oxalate, and other iron coordination compounds used in analytical and photographic applications.
Applications of Ferric Ammonium Oxalate
Photographic Processes
- Used in alternative photographic printing (cyanotype)
- Applied in making blueprints and architectural drawings
- Used in photographic sensitizers
Analytical Chemistry
- Used as an analytical reagent for detecting various ions
- Applied in spectrophotometric determinations
- Used in developing analytical methods
Industrial Applications
- Used in electroplating baths
- Applied to the metal surface treatment
Safety & Handling Guidelines
- Avoid inhalation of dust
- Use gloves and protective clothing
- Handle in well-ventilated areas
- Store in tightly closed containers protected from light
- Keep away from strong acids and bases
- Follow COA and MSDS instructions strictly
Where to Buy Ferric Ammonium Oxalate?
Ferric Ammonium Oxalate Manufacturer
ChemicalBull supplies high-purity Ferric Ammonium Oxalate for photographic and analytical applications.
Ferric Ammonium Oxalate Supplier & Distributor
- Available in various pack sizes for specialized use
- High-purity grades for analytical and photographic applications
- Protected packaging (light-resistant)
- COA, MSDS provided
MSDS
Includes detailed safety instructions and handling protocols.
Frequently Asked Questions (FAQs)
-
What is Ferric Ammonium Oxalate used for?
Used in cyanotype photography, blueprint processes, analytical chemistry, and as a photosensitive reagent. -
What is the molecular formula?
Complex formula: (NH₄)₃[Fe(C₂O₄)₃]·nH₂O with varying hydration. -
How is it used in blueprints?
Light-sensitive iron compound; UV exposure reduces Fe(III) to Fe(II), producing Prussian blue when developed.