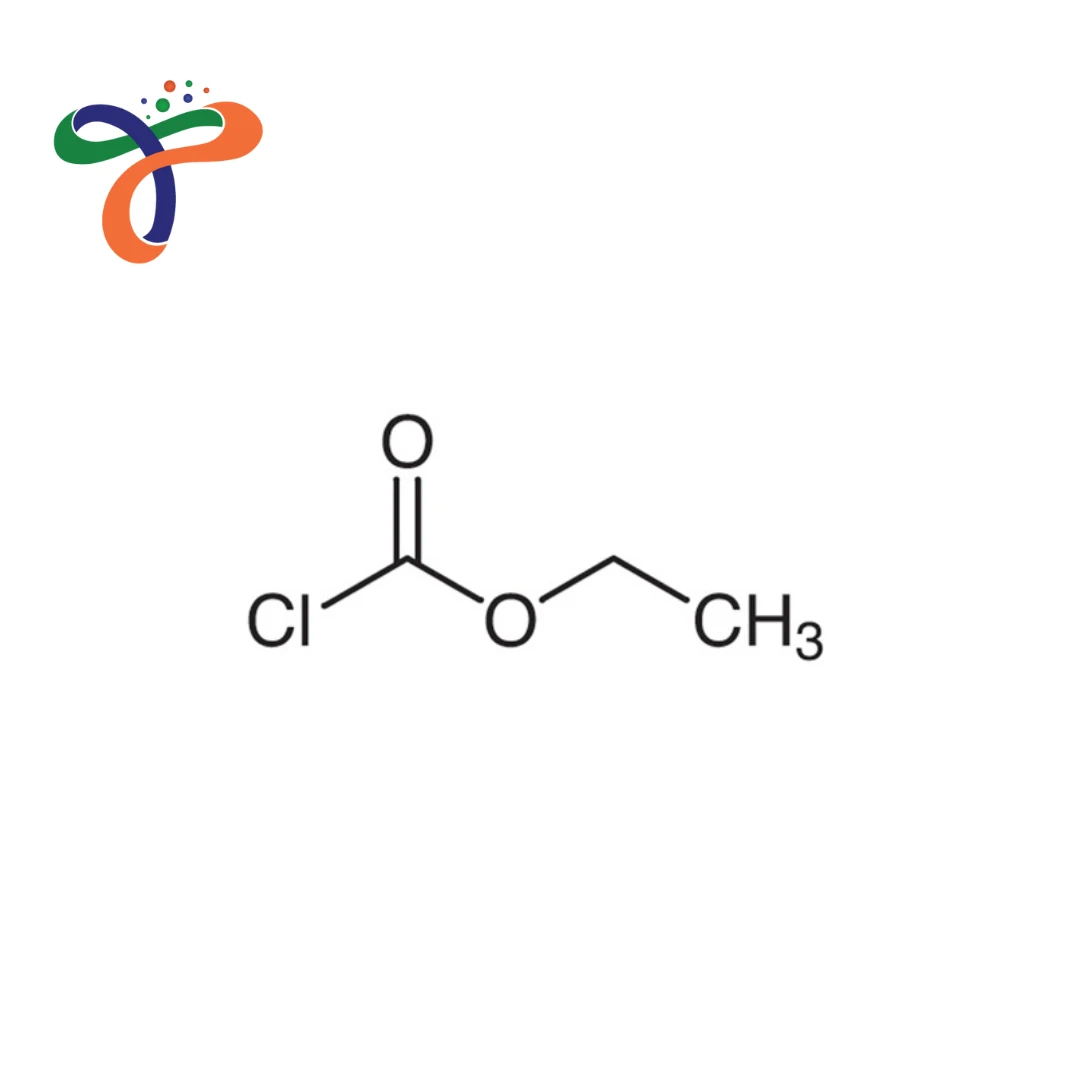
Ethyl Chloroformate | 541-41-3
Cas No :
541-41-3
Synonyms :
Form :
Liquid
Molecular Weight :
108.52
Molecular Formula :
C3H5CLO2
Boiling Point :
93 °C
Melting Point :
81 °C
Flash Point :
16 °C
Solubility :
Chloroform (Soluble), Ethyl Acetate (Slightly)
Description :
An Overview of Ethyl Chloroformate
Ethyl Chloroformate is an extremely reactive organic compound widely used as an acylating chemical reagent for pharmaceutical synthesis, as well as peptide chemistry and organic transformations. It is renowned for its effectiveness in activating carboxylic acids, making mixed anhydrides, and creating the ethoxycarbonyl group. Ethyl Chloroformate is extensively used in the manufacturing of pharmaceuticals and organic chemistry, as well as in fine chemical synthesis.
It is a colourless, pale yellow-coloured liquid that has a strong, offensive, and lachrymatory scent. The chloroformate esters structure offers an excellent reaction with nucleophiles, which include alcohols, amines and carboxylates, in mild conditions.
The products that are related include Methyl chloroformate, Benzyl chloroformate, Isobutyl Chloroformate, and other reagents of chloroformate used in the synthesis of pharmaceuticals, as well as protecting groups in chemistry.
Applications of Ethyl Chloroformate
Pharmaceutical Synthesis
- Utilised to activate carboxylic acids to initiate coupling reactions
- Used to peptide bonding using the mixed anhydride technique
- Utilised to introduce Ethoxycarbonyl (Cbz), safeguarding the groups
- Used in the production of intermediates for pharmaceutical production.
- It is used in the preparation of the carbamate-containing drugs
- Used in the synthesis of prodrugs as well as modification
- It is used in the synthesis of amino acid derivatives
- Used in the manufacture of antibiotics as well as other APIs
Organic Synthesis
- It is used as part of the Einhorn mixed anhydride method. Einhorn mixed anhydride technique
- It is used to make carbamates by combining amines
- Utilised for the esterification of alcohols
- It is used in the process of synthesising carbonates
- used in Schotten-Baumann reactions
- Used to protect group creation and elimination
- Utilised in the complete synthetic process of natural ingredients
- Applied in developing new synthetic methodologies
Fine Chemical Manufacturing
- It is used to make agricultural chemical intermediates
- Used in the manufacturing of colours and dyes
- Utilised in synthesising speciality chemicals
- Utilised in the manufacture of the performance chemical
- Used in developing new materials
- Used in the preparation of analytic standards
Safety and Handbook Guidelines for Handling
- HIGHLY CORROSIVE, LACHRYMATORY and highly toxic--handle extremely cautiously
- Rapidly reacts with water, dispersing CO2 and HCl.
- Utilise only within a fume hood that is well ventilated and with a suitable containment
- Wear full PPE, including acid-resistant gloves, face shield, and lab coat
- Storage in containers that are tightly sealed under cool, dry, and anhydrous conditions.
- Stay clear of bases, moisture, water and alcohols
- Make sure you have an emergency shower and eyewash readily available
- Be disposed of in accordance with the rules for hazardous waste
- Use the COA as well as the MSDS guidelines strictly
Where to Buy Ethyl Chloroformate?
Ethyl Chloroformate Manufacturer
ChemicalBull supplies high-purity Ethyl Chloroformate for pharmaceutical and research applications with appropriate safety packaging.
Ethyl Chloroformate Supplier & Distributor
- Available in controlled quantities for pharmaceutical and research use
- High-purity anhydrous grade (97%, 99%)
- Special moisture-resistant packaging provided
- COA, MSDS, and comprehensive safety documentation provided
MSDS
Includes comprehensive safety instructions, reactivity information, emergency procedures, and first aid measures.
Frequently Asked Questions (FAQs)
-
What is Ethyl Chloroformate used for?
Used as an acylating reagent in pharmaceutical synthesis for peptide coupling, carbamate synthesis, carboxylic acid activation, and protecting group chemistry. -
What is the chemical formula of Ethyl Chloroformate?
Molecular formula: C₃H₅ClO₂ or ClCOOC₂H₅ with molecular weight 108.52 g/mol. -
What is the structure of Ethyl Chloroformate?
Contains ethyl ester group attached to carbonyl chloride; systematic name: ethyl carbonochloridate.