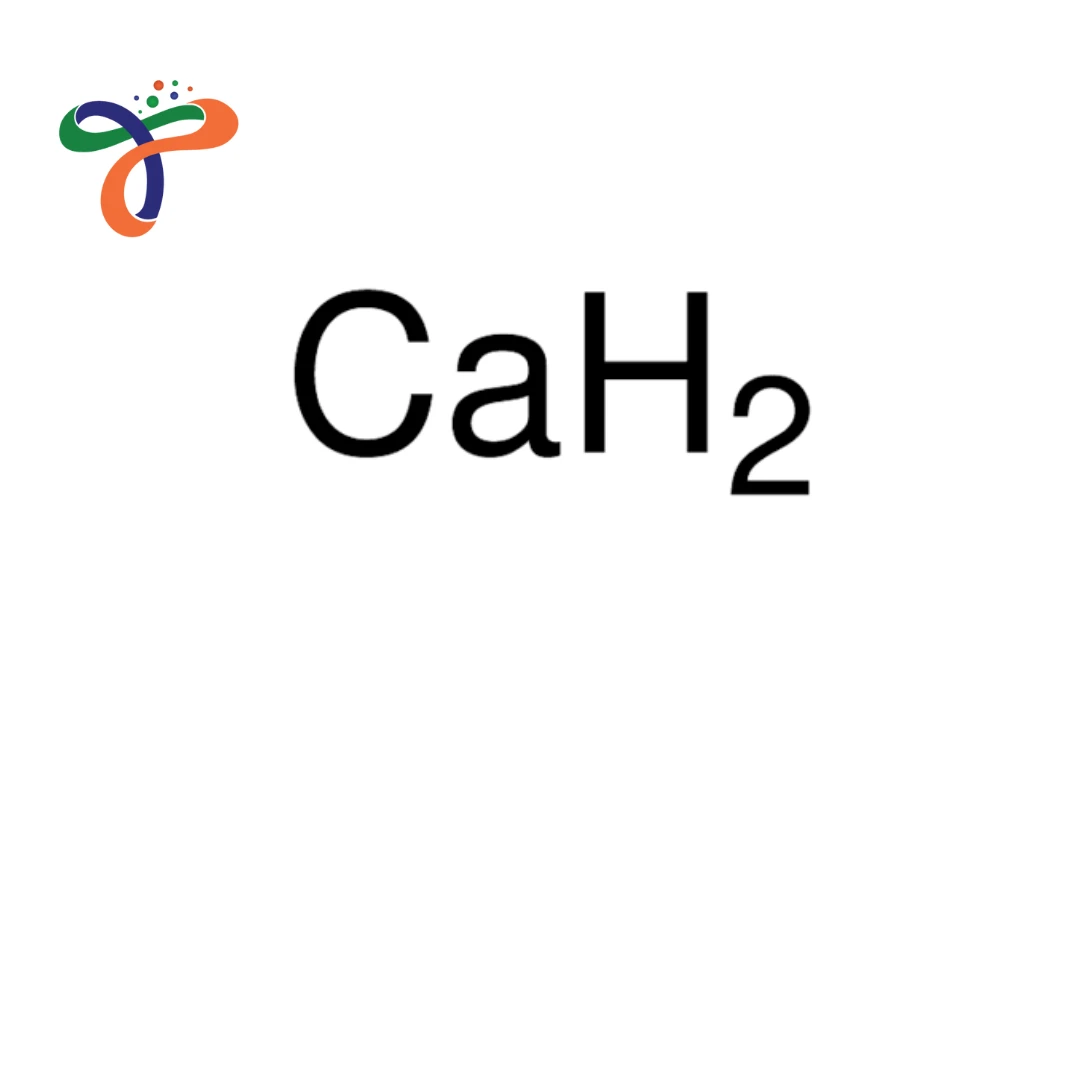Calcium Hydride
Form :
Powder
Molecular Weight :
42.094 G/Mol
Boiling Point :
900 °C
Melting Point :
850 °C
Flash Point :
500 °C
Solubility :
Insoluble In Water; Reacts With Water To Form Calcium Hydroxide And Hydrogen Gas
Description :
Overview of Calcium Hydride
Calcium Hydride is a reducing agent and desiccant used for drying solvents, generating hydrogen gas, and as a chemical reagent. Known for its powerful water-scavenging properties, ability to remove trace moisture from organic solvents, and use as a hydrogen source, Calcium Hydride is widely used in organic chemistry laboratories, pharmaceutical synthesis, and specialty chemical production.
This compound appears as a gray to white powder. Its metal hydride structure provides powerful reducing and drying capability.
Related products include Sodium Hydride, Lithium Aluminum Hydride, and other metal hydrides used in organic synthesis and drying applications.
Applications of Calcium Hydride
Solvent Drying
- Used for removing water from organic solvents
- Applied in preparing anhydrous solvents
- Used in drying ethers, hydrocarbons, and amines
Pharmaceutical Synthesis
- Used in moisture-sensitive reactions
- Applied in pharmaceutical intermediate production
- Used in anhydrous synthesis procedures
- Applied in medicinal chemistry research
Industrial Applications
- Used as a hydrogen generation source (portable)
- Applied in reducing agent applications
Safety & Handling Guidelines
- REACTS VIOLENTLY WITH WATER—releases hydrogen gas
- FLAMMABLE—hydrogen generation creates a fire hazard
- Use only under strictly anhydrous conditions
- Handle in a well-ventilated fume hood or an inert atmosphere
- Wear appropriate PPE, including gloves, goggles, and protective clothing
- Store in tightly sealed containers under inert gas
Where to Buy Calcium Hydride?
Calcium Hydride Manufacturer
ChemicalBull supplies high-purity Calcium Hydride for pharmaceutical and research applications.
Calcium Hydride Supplier & Distributor
- Available in bulk quantities for industrial and research use
- High-purity anhydrous grade (93-95% CaH₂)
- Special moisture-resistant packaging under inert gas
- COA, MSDS, and comprehensive safety documentation provided
MSDS
Includes comprehensive reactivity warnings, fire hazard information, and handling protocols.
Frequently Asked Questions (FAQs)
-
What is Calcium Hydride used for?
Used primarily for drying organic solvents, generating hydrogen gas, and as a reducing agent in anhydrous chemistry. -
What is the molecular formula?
Molecular formula: CaH₂ with molecular weight 42.09 g/mol. -
How does it dry solvents?
Reacts with trace water, forming calcium hydroxide and releasing hydrogen; removes moisture, producing ultra-dry solvents.