Trichloroethane | 71-55-6
Cas No :
71-55-6
Form :
Liquid
Molecular Weight :
133.40 G/Mol
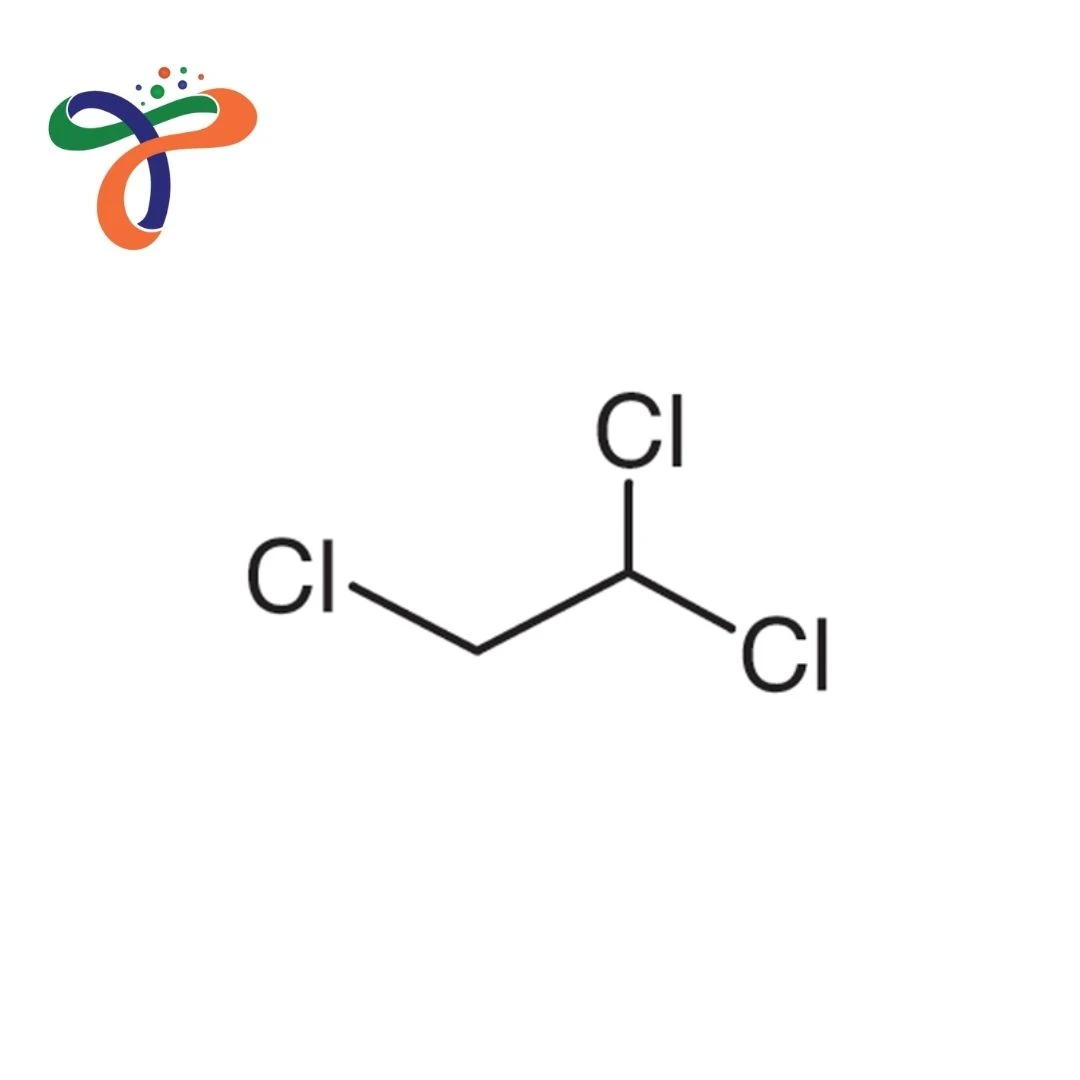
Molecular Formula :
C2H3CL3
Boiling Point :
74 °C
Melting Point :
-76 °C
Flash Point :
-20 °C
Solubility :
Not Soluble In Water; Soluble In Organic Solvents
Description :
Overview of Trichloroethane
Trichloroethane is a chlorinated hydrocarbon that exists in two isomeric forms: 1,1,1-Trichloroethane and 1,1,2-Trichloroethane, historically used as industrial solvents, degreasers, and chemical intermediates. Known for their excellent solvent properties for oils, greases, and organic materials, these compounds were widely used in industrial cleaning, though their use is now heavily restricted due to ozone depletion potential and health concerns.
The two isomers are transparent liquids that have a distinctive flavor that is sweet and has a similar odor to chloroform. The chlorinated structure of the isomers has excellent cleaning properties, but issues with health and the environment caused a phase-out in international conventions.
Similar products are Perchloroethylene, Trichloroethylene, and other chlorinated solvents, with restricted or controlled usage.
Applications of Trichloroethane
The Industrial History of the Past (1,1,1-TCE)
- The substance was previously utilized to degrease metals (largely eliminated)
- Used for cold cleaning and degreasing vapors (discontinued throughout the majority of regions)
- It was used for a long time in electronic cleaning (replaced with alternative methods)
- Used in the formulation of aerosol propellants (banned by the Montreal Protocol)
- Utilized in adhesives as well as coating formulas (restricted)
- It is used in dry-cleaning operations (discontinued)
Limited Current Applications
- Limited use for certain special industrial techniques under the terms of permits
- Utilized in laboratory-controlled research
- It is used in analytical chemistry applications (reducing the use)
- Used in the process of an intermediate pharmaceutical synthesis (limited, however, under control)
- It is used to make various other chemicals that are subject to strict regulations.
- It is used in some formulas of solvent mixtures, with limitations.
Chemical Synthesis (1,1,2-TCE)
- Limited use as a chemical intermediate
- Applied in manufacturing vinylidene chloride (restricted)
- Used in producing other chlorinated compounds
- Applied in controlled research applications
- Subject to strict regulatory controls
Safety & Handling Guidelines
- TOXIC and OZONE DEPLETING—heavily regulated substance
- Avoid inhalation of vapors—causes CNS depression
- Use only with appropriate authorization and permits
- Handle in a well-ventilated fume hood with proper controls
- Wear appropriate PPE, including gloves, goggles, and protective clothing
- Store in tightly sealed containers in approved storage areas
Where to Buy Trichloroethane?
Trichloroethane Manufacturer
ChemicalBull supplies Trichloroethane for authorized applications only, with comprehensive regulatory compliance and documentation.
Trichloroethane Supplier & Distributor
- Available in very limited quantities for authorized use only
- Requires appropriate permits and documentation in most jurisdictions
- Subject to Montreal Protocol reporting requirements
- International shipping is highly restricted or prohibited
- COA, MSDS, and regulatory compliance documentation provided
- Alternative solvents recommended for most applications
MSDS
Includes comprehensive toxicity warnings, ozone depletion information, environmental hazard data, and strict handling protocols.
Frequently Asked Questions (FAQs)
-
What is Trichloroethane used for?
Historically used as an industrial degreasing solvent and aerosol propellant; now heavily restricted with very limited authorized uses due to ozone depletion and health concerns. -
What is the difference between 1,1,1 and 1,1,2 isomers?
1,1,1-Trichloroethane: three chlorines on the same carbon, less toxic, major ozone depleter (phased out under Montreal Protocol); 1,1,2-Trichloroethane: chlorines on adjacent carbons, more toxic, not ozone depleting but carcinogenic. -
Why was 1,1,1-Trichloroethane banned?
Class I ozone-depleting substance under the Montreal Protocol; production and consumption phased out in developed countries (1996) and developing countries (2015) except for essential uses.