Ammonium Dichromate | 05-09-7789
Cas No :
05-09-7789
Form :
Solid
Molecular Weight :
252.07 G/Mol
Molecular Formula :
CR2H2O7.2H3N
Boiling Point :
200 °C
Melting Point :
195 °C
Flash Point :
200 °C
Solubility :
Soluble In Water
Description :
Overview of Ammonium Dichromate
Ammonium Dichromate is an organic chemical that is used for chromium in a variety of chemical applications in the industrial sector, as well as analytical and photography. The compound is known for its formidable characteristics in oxidation, and its distinct colour of orange-red. Ammonium Dichromate is used for manufacturing, lab demonstrations and in the development of specialised chemical techniques.
The compound is a red-orange crystalline solid that has distinctive features. Its distinctive oxidising nature, as well as the chromium content, makes it useful for specific research and industrial uses, even though the health and environmental aspects restrict some of its traditional uses.
Other related products include Dichromate, Ammonium Iodide, Chromic Acid, as well as various other chromium compounds that are utilised in industrial and analytical uses. As with Calcium Iodide, it requires cautious handling due to its chemical characteristics.
Applications of Ammonium Dichromate
Industrial Applications
- It is used in tanning leather (chromium tanning)
- It is used in dyeing textiles and printing
- Utilised in treatments to preserve wood
- Utilised in the production of pigments
- It is used for corrosion prevention.
- Applied in certain electroplating processes
Analytical & Laboratory
- Utilised in the analysis of Chemistry and the titration process.
- Used in organic syntheses
- It is used to demonstrate the effects of the "volcano" reaction (decomposition)
- Used in microscopy as well as histology staining
- Utilised in photography and lithography
- Applications for research in the chemistry of chromium
Manufacturing & Synthesis
- It is used in the production of other chromium compounds
- Used in the preparation of catalysts
- It is used for the oxidation process of alcohols as well as other organic compounds.
- It is used in specialist chemical syntheses.
Safety & Handling Guidelines
- HIGHLY toxic and CARCINOGENIC (hexavalent Chromium)
- Use only in hazardous chemical zones.
- Wear appropriate PPE, including a respirator, gloves, and goggles
- Manage inside a fume hood using the proper protection
- A strong oxidiser. Keep away from the combustibles
- Keep tightly shut containers, secured storage
- Be disposed of in accordance with the rules for hazardous waste
- Be sure to follow COA and MSDS guidelines completely.
Where to Buy Ammonium Dichromate?
Ammonium Dichromate Manufacturer
ChemicalBull supplies analytical-grade Ammonium Dichromate for authorisedauthorised industrial and research applications, ensuring strict regulatory compliance.
Ammonium Dichromate Supplier & Distributor
- Available in controlled quantities for licensed facilities
- Requires proper authorisation and safety documentation
- Analytical and technical grades
- COA, MSDS, and comprehensive safety information provided
MSDS
Includes comprehensive safety instructions, carcinogenicity warnings, exposure controls, and emergency procedures.
Frequently Asked Questions (FAQs)
-
What are the Ammonium Dichromate buy restrictions?
Subject to regulations due to toxicity and environmental concerns, licenses may be required and are increasingly restricted in many regions. -
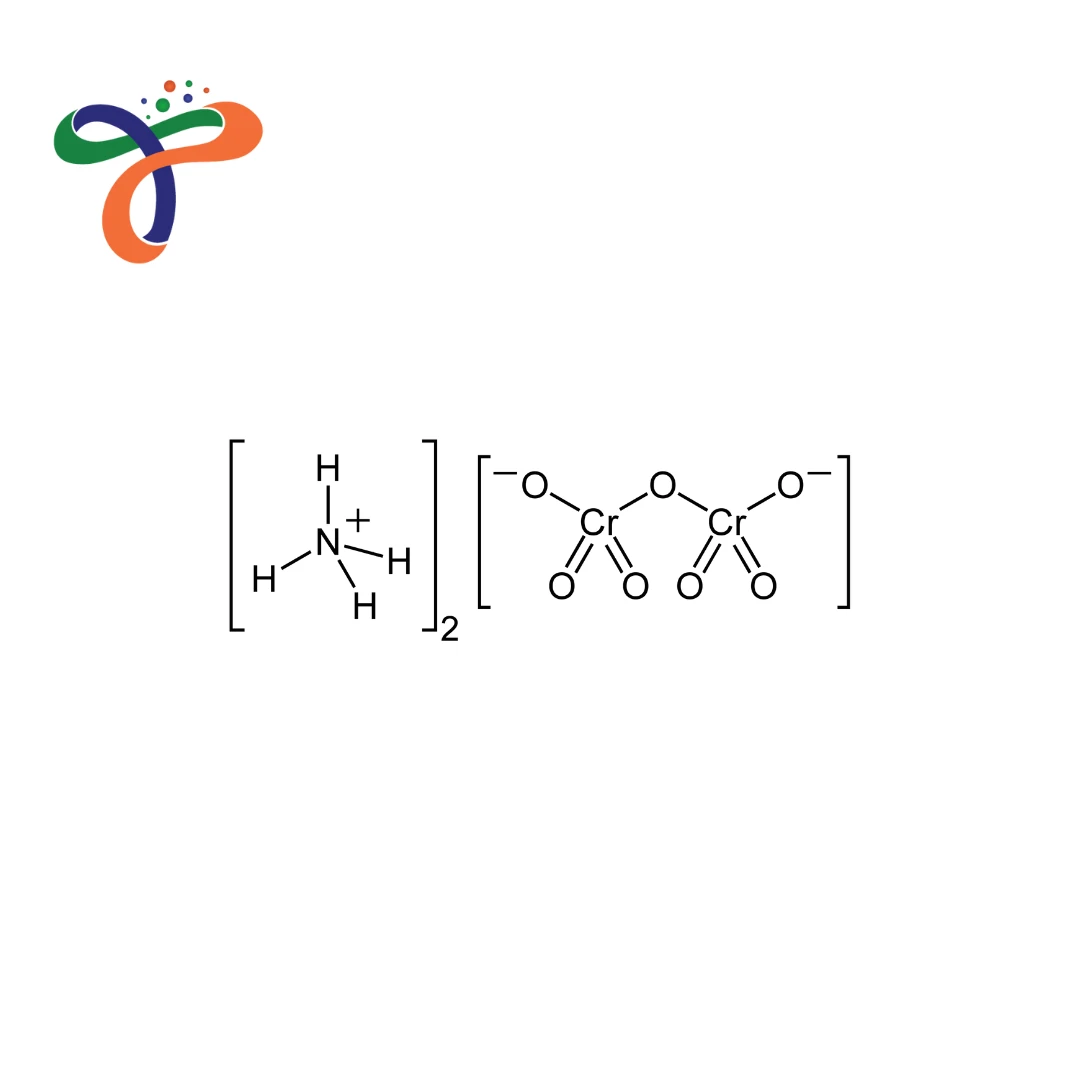
What is the Ammonium Dichromate structure?
Contains dichromate anion (Cr₂O₇²⁻) with two ammonium cations; molecular formula: (NH₄)₂Cr₂O₇. -
What is Ammonium Dichromate formula?
Chemical formula: (NH₄)₂Cr₂O₇ with molecular weight 252.06 g/mol. -
What is the colour of Ammonium Dichromate?
Distinctive orange-red to reddish-orange crystalline solid. -
What is Ammonium Dichromate used for?
Used in analytical chemistry, leather tanning, pigment manufacturing, organic oxidations, and specialised industrial applications.