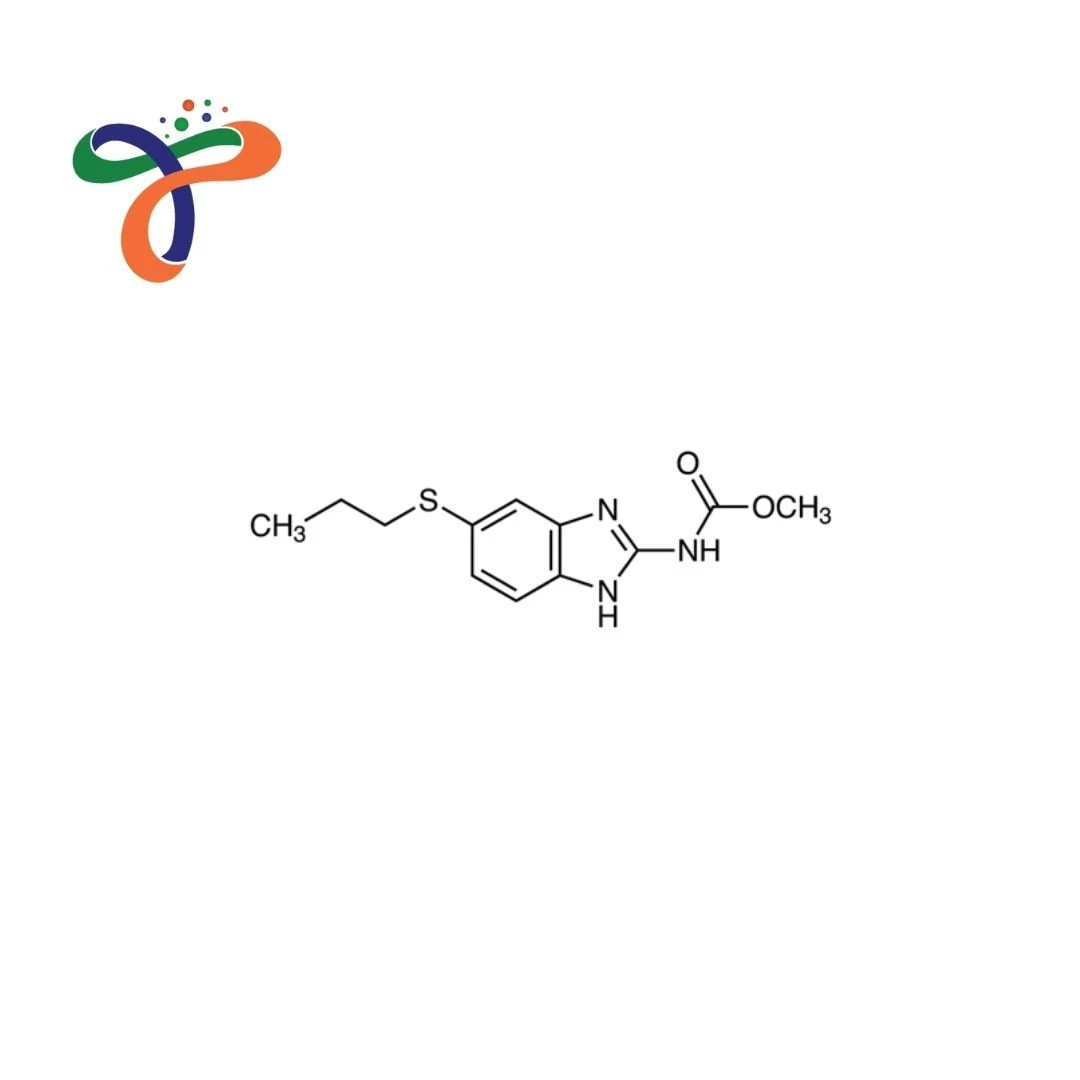
Albendazole | 54965-21-8
Cas No :
54965-21-8
Form :
Solid
Molecular Weight :
265.33 G/Mol
Molecular Formula :
C12H15N3O2S
Boiling Point :
190 °C
Melting Point :
173 °C
Flash Point :
190 °C
Solubility :
Poorly Soluble In Water; Soluble In Organic Solvents Such As Acetone And Ethanol
Description :
Overview of Albendazole
Albendazole is a broad-spectrum anthelmintic pharmaceutical active ingredient widely used in the treatment of parasitic worm infections. It belongs to the benzimidazole class of compounds and works by inhibiting tubulin polymerisation in parasites, leading to impaired glucose uptake and eventual parasite death.
Albendazole appears as a white to off-white crystalline powder and is practically insoluble in water but soluble in organic solvents. It exhibits high efficacy at low doses and is commonly formulated into tablets, chewable tablets, and oral suspensions. Due to its effectiveness against a wide range of intestinal and tissue parasites, Albendazole is extensively used in human and veterinary pharmaceutical formulations.
ChemicalBull supplies pharma-grade Albendazole with high purity, controlled particle size, and consistent batch-to-batch quality suitable for regulated pharmaceutical manufacturing. Each shipment is supported with complete documentation, including MSDS, COA, and technical data sheets, ensuring compliance with pharmaceutical industry standards.
As a trusted chemical supplier and reliable distributor, ChemicalBull ensures dependable availability of Albendazole for pharmaceutical manufacturers. You may also like Mebendazole, Ivermectin, Praziquantel, and Pharmaceutical APIs for related antiparasitic applications.
Applications of Albendazole
Pharmaceutical Formulations
-
Used in tablets, chewable tablets, and oral suspensions
-
Effective against intestinal and tissue parasitic infections
-
Applied in single-dose and multi-dose treatment regimens
Hospital & Clinical Use
-
Used in the treatment of helminth infections
-
Applied in mass deworming and public health programs
-
Widely prescribed in human healthcare
Veterinary Applications
-
Used in antiparasitic formulations for animals
-
Effective against a broad range of parasitic worms
-
Applied in livestock and companion animal treatments
Pharmaceutical Research & Development
-
Used as a reference standard in antiparasitic research
-
Applied in formulation development and bioavailability studies
-
Suitable for controlled pharmaceutical laboratories
Safety & Handling Guidelines
-
Handle using gloves and appropriate protective equipment
-
Avoid inhalation of powder
-
Use in well-ventilated pharmaceutical facilities
-
Store in tightly sealed containers protected from light and moisture
-
Follow MSDS and standard pharmaceutical safety practices
Where to Buy Albendazole?
Albendazole Manufacturer
ChemicalBull supplies pharmaceutical-grade Albendazole suitable for antiparasitic drug formulations and regulated pharmaceutical applications.
Albendazole Supplier & Distributor
-
Bulk and customised pharmaceutical packaging options
-
Export-grade quality with COA, TDS & MSDS
-
Reliable supply across India and international markets
-
Trusted chemical supplier for pharmaceutical APIs
Albendazole MSDS
The Albendazole MSDS provides essential information on safe handling, storage conditions, exposure controls, stability, and emergency procedures. Always review the MSDS before bulk handling or pharmaceutical manufacturing use.
Frequently Asked Questions (FAQs)
-
What is Albendazole mainly used for?
Albendazole is mainly used for the treatment of parasitic worm infections. -
Is Albendazole suitable for pharmaceutical manufacturing?
Yes, pharma-grade Albendazole is widely used in regulated pharmaceutical production. -
Is Albendazole water-soluble?
No, it is practically insoluble in water but soluble in organic solvents. -
Can Albendazole be used in veterinary medicines?
Yes, it is commonly used in both human and veterinary antiparasitic formulations. -
Is safety documentation provided with the supply?
Yes, every shipment includes MSDS, COA, and complete technical documentation.