2-Amino-3,5-Dibromobenzaldehyde | 50910-55-9
Cas No :
50910-55-9
Synonyms :
Form :
Solid
Molecular Weight :
278.93 G/Mol
Molecular Formula :
C7H5BR2NO
Boiling Point :
260 °C
Melting Point :
128 °C
Flash Point :
92 °C
Solubility :
Slightly Soluble In Water; Soluble In Organic Solvents Such As Acetone, Dichloromethane, And Ethanol
Description :
Overview of 2-Amino-3,5-Dibromobenzaldehyde
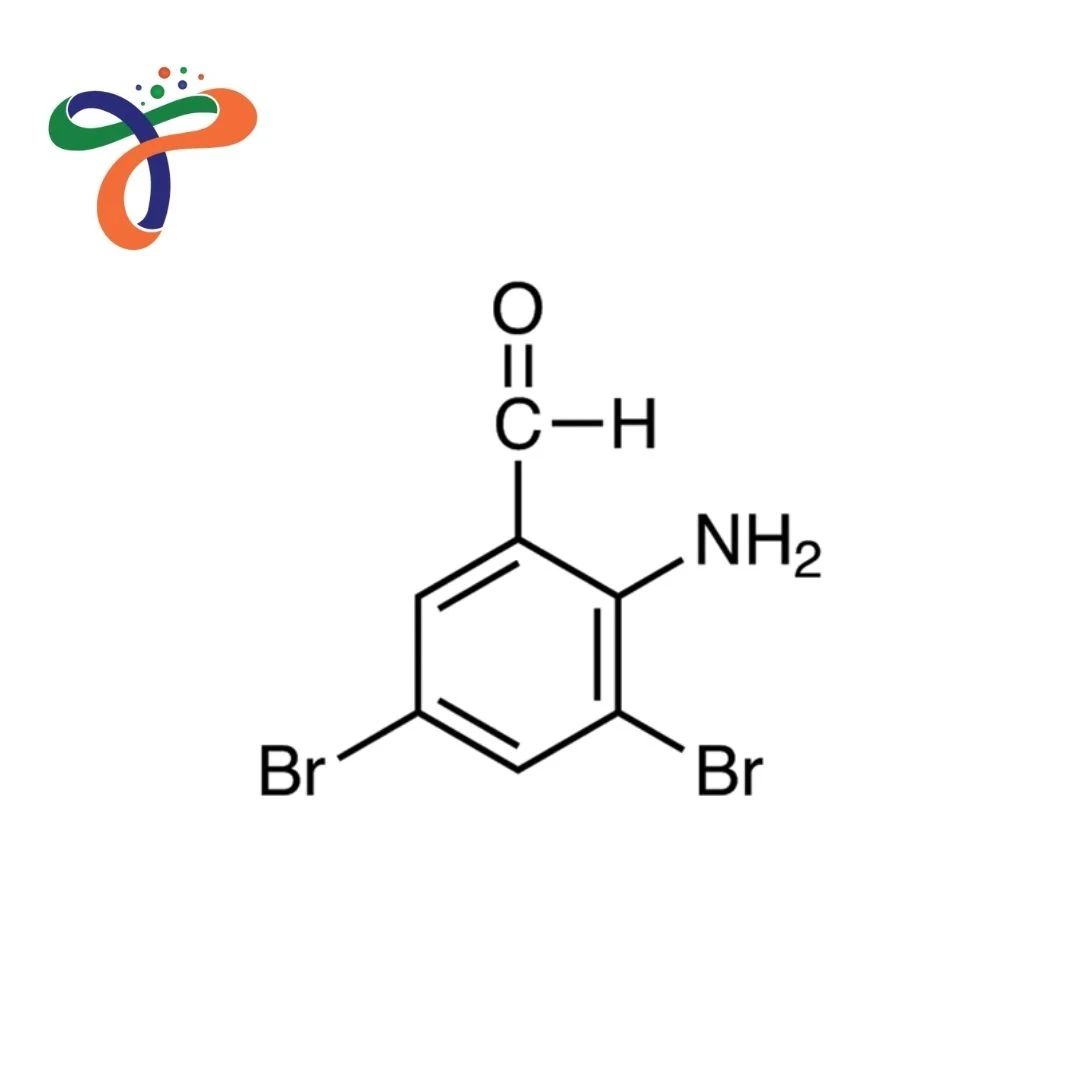
2-Amino-3,5-Dibromobenzaldehyde is a specialised industrial halogenated aromatic intermediate, typically supplied as a light yellow to pale brown crystalline solid. It contains an aldehyde (-CHO) group and an amino (-NH₂) group on a benzene ring substituted with two bromine atoms at the 3 and 5 positions, providing high reactivity for further functionalisation.
As an industrial chemical, 2-Amino-3,5-Dibromobenzaldehyde is widely used in organic synthesis, heterocyclic compound development, dye and pigment intermediates, and specialty chemical manufacturing, where controlled purity and precise substitution patterns are essential.
Applications of 2-Amino-3,5-Dibromobenzaldehyde
Organic Synthesis & Functional Intermediates
2-Amino-3,5-Dibromobenzaldehyde is widely used as a key building block in multi-step synthesis reactions, particularly for Schiff base formation, condensation, and cyclisation processes. It is commonly processed alongside Aniline, Thionyl Chloride, and Acetic Anhydride in structured industrial synthesis systems.
Heterocyclic & Advanced Compound Development
Due to the presence of both aldehyde and amino groups, this compound supports heterocyclic ring formation and brominated aromatic derivative production, enhancing reactivity and downstream modification potential. It is often evaluated with Palladium on Activated Carbon 5% in controlled coupling reactions.
Specialty Chemical & Dye Intermediate Manufacturing
2-Amino-3,5-Dibromobenzaldehyde is applied in specialty dye, pigment, and performance chemical manufacturing, where halogenated aromatic aldehydes provide improved chemical stability and selectivity.
Safety & Handling Guidelines
-
Store in a cool, dry, and well-ventilated area
-
Keep containers tightly sealed and clearly labelled
-
Avoid inhalation of dust and direct contact with skin or eyes
-
Use protective gloves, goggles, and suitable industrial clothing
-
Keep away from strong oxidising and reducing agents
-
Handle under controlled industrial conditions
-
Dispose of waste and packaging according to regulatory guidelines
Where to Buy 2-Amino-3,5-Dibromobenzaldehyde?
2-Amino-3,5-Dibromobenzaldehyde Manufacturer
2-Amino-3,5-Dibromobenzaldehyde is manufactured for industrial-grade applications requiring controlled bromine content, stable aldehyde functionality, and reliable performance in synthesis industries.
2-Amino-3,5-Dibromobenzaldehyde Supplier & Distributor
2-Amino-3,5-Dibromobenzaldehyde is supplied in bulk and industrial packaging formats for manufacturers requiring dependable quality and regulated handling. Buy 2-Amino-3,5-Dibromobenzaldehyde from Chemicalbull Pvt. Ltd., a trusted industrial chemicals supplier and distributor in India, supporting customers with reliable sourcing and compliant logistics.
MSDS for 2-Amino-3,5-Dibromobenzaldehyde
The MSDS for 2-Amino-3,5-Dibromobenzaldehyde provides detailed information on hazards, safe handling practices, storage conditions, exposure controls, and emergency response procedures. Always review the MSDS before industrial use.
Frequently Asked Questions (FAQs)
-
What is the structure of 2-Amino-3,5-Dibromobenzaldehyde?
It consists of a benzene ring substituted with an amino (-NH₂) group at position 2, bromine atoms at positions 3 and 5, and an aldehyde (-CHO) group attached to the aromatic ring. -
What is 2-Amino-3,5-Dibromobenzaldehyde used for?
It is used in organic synthesis, heterocyclic compound formation, specialty intermediate manufacturing, and dye and pigment production. -
How is 2-Amino-3,5-Dibromobenzaldehyde synthesised?
It is typically synthesised through controlled bromination and functional group transformation of substituted benzaldehyde derivatives under industrial reaction conditions.