Overview of 1,4-Difluorobenzene
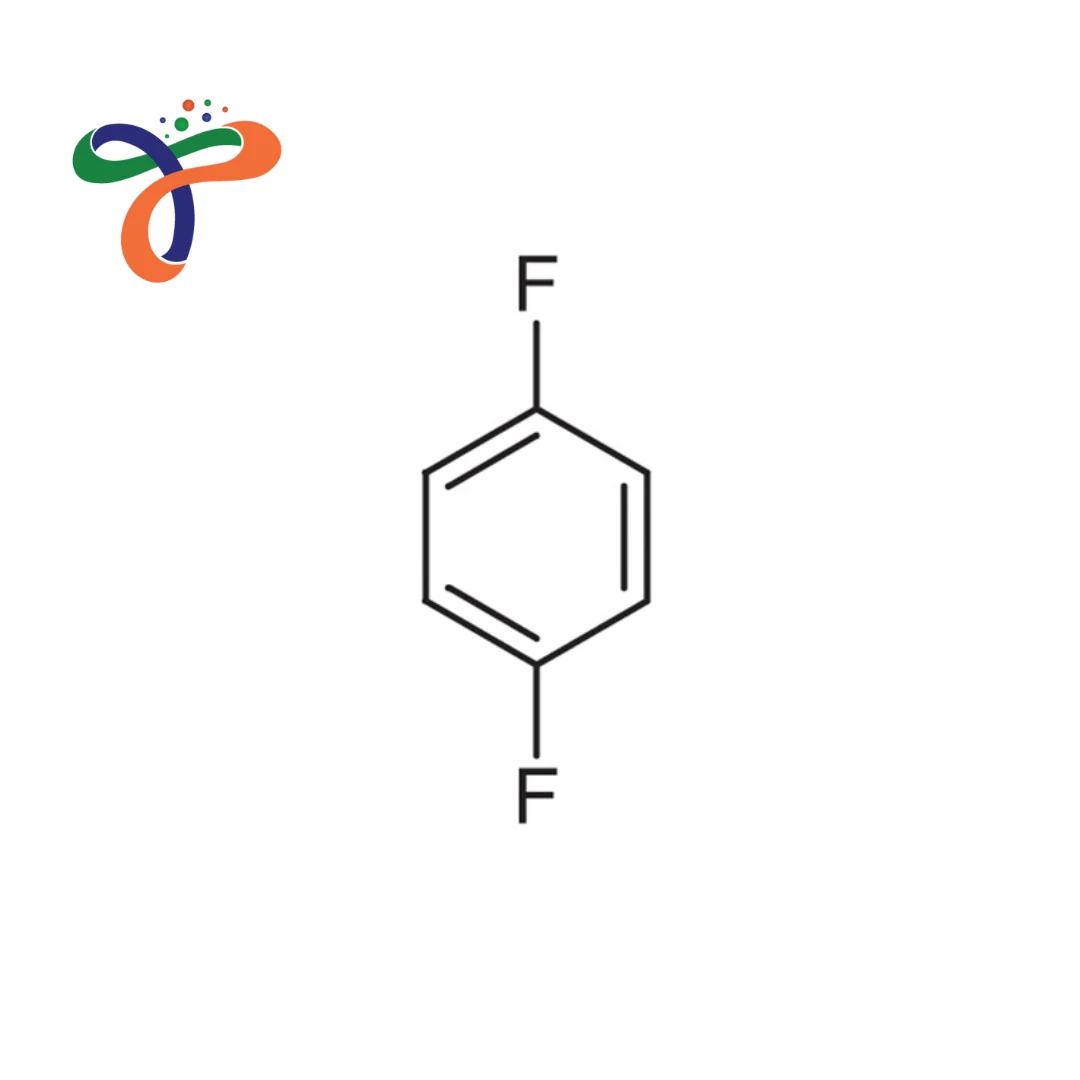
1,4-Difluorobenzene, also known as p-difluorobenzene, is an aromatic halogenated compound widely used in chemical synthesis, pharmaceutical intermediates, and specialty material production. It consists of a benzene ring with two fluorine atoms attached at the para (1,4) positions, giving the compound a symmetrical molecular arrangement. The 1,4-difluorobenzene formula is C₆H₄F₂, and the 1,4-difluorobenzene structure features fluorine atoms positioned opposite each other on the aromatic ring.
This compound is valued for its chemical stability and controlled reactivity in fluorinated organic synthesis. The 1,4-difluorobenzene boiling point is approximately 88–90°C, while the density of 1,4-difluorobenzene is about 1.19 g/cm³ at 25°C. High-quality high-purity p-difluorobenzene is commonly used in industrial production and research laboratories where consistent performance and purity are required.
Applications of 1,4-Difluorobenzene
Pharmaceutical & Chemical Intermediates
p-Difluorobenzene is widely used as a starting material in pharmaceutical synthesis and specialty organic chemistry. The presence of fluorine atoms enhances molecular stability and helps create fluorinated intermediates used in drug development.
It may be processed in reactions involving Potassium Carbonate, Copper Catalysts, and other reagents for fluorinated compound synthesis.
Agrochemical Production
Fluorinated aromatic compounds are frequently used in agrochemical formulations. This material serves as a building block for producing intermediates required for pesticides, herbicides, and crop protection chemicals.
Polymer & Specialty Material Manufacturing
In advanced material science, fluorinated benzene derivatives contribute to the production of high-performance polymers and specialty coatings with improved chemical resistance and durability.
Laboratory Research & Organic Synthesis
Researchers utilize this compound in studies involving aromatic substitution reactions, fluorination chemistry, and the preparation of complex fluorinated molecules.
Safety & Handling Guidelines
-
Store in sealed containers in a cool, dry environment
-
Keep away from ignition sources and incompatible chemicals
-
Avoid inhalation of vapors and prolonged exposure
-
Use protective gloves, goggles, and appropriate protective clothing
-
Ensure proper ventilation when handling the material
-
Follow environmental and chemical safety regulations for disposal
Always review the official safety documentation before handling or processing.
Where to Buy 1,4-Difluorobenzene?
1,4-Difluorobenzene Manufacturer
This fluorinated aromatic compound is produced using controlled fluorination processes to ensure high purity and consistent chemical composition suitable for industrial and research applications.
1,4-Difluorobenzene Supplier & Distributor
Chemicalbull Pvt. Ltd. supplies high-purity p-difluorobenzene for chemical manufacturers, laboratories, and specialty chemical industries. The company offers reliable bulk distribution and quality-controlled supply.
For information about 1 4 difluorobenzene price, bulk availability, or product specifications, contact Chemicalbull Pvt. Ltd. for the latest quotation.
MSDS for 1,4-Difluorobenzene
The MSDS contains important safety information including hazard identification, exposure control measures, storage conditions, and emergency response procedures. Reviewing the safety data sheet is recommended before use.
Frequently Asked Questions (FAQs)
-
What is the 1 4 difluorobenzene formula?
The molecular formula is C₆H₄F₂.
-
What is the 1 4 difluorobenzene structure?
It consists of a benzene ring with fluorine atoms attached at the para (1,4) positions.
-
What is the boiling point of this compound?
The boiling point is approximately 88–90°C.
-
What is the density of 1 4 Difluorobenzene?
Its density is around 1.19 g/cm³ at 25°C.
-
1 4-Difluorobenzene vs. 1 2-Difluorobenzene – what is the difference?
The difference lies in the position of fluorine atoms on the benzene ring. In the para isomer they are opposite each other, while in the ortho isomer they are adjacent.
-
What is the price of 1 4 Difluorobenzene?
Pricing depends on purity level and order quantity. Contact Chemicalbull Pvt. Ltd. for current bulk supply details.