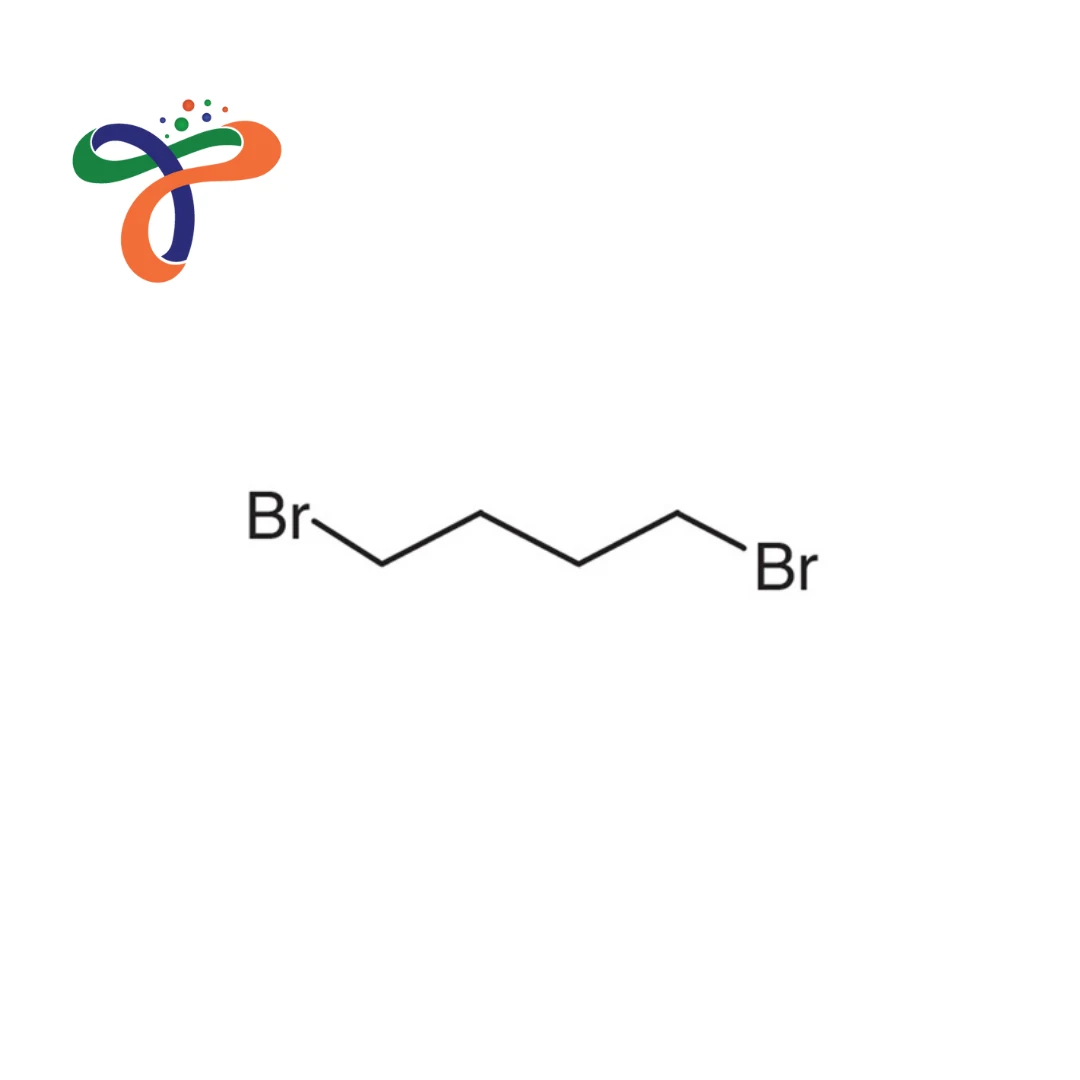
1,4-Dibromobutane | 110-52-1
Cas No :
110-52-1
Synonyms :
Form :
Liquid
Molecular Weight :
213.90 G/Mol
Molecular Formula :
C4H8BR2
Boiling Point :
230 °C
Melting Point :
-30 °C
Flash Point :
120 °C
Solubility :
Insoluble In Water; Soluble In Organic Solvents Such As Dichloromethane And Chloroform
Description :
Overview of 1,4-Dibromobutane
1,4-Dibromobutane (CAS: 110-52-1) is a di-halogenated aliphatic compound widely used in organic synthesis, polymer chemistry, pharmaceutical intermediates, and specialty chemical production. It appears as a colorless to pale yellow liquid and is valued for its high reactivity in nucleophilic substitution, cyclization, and cross-linking reactions.
This compound is commonly employed as a building block for heterocyclic compounds, polymer cross-linkers, and fine chemical intermediates. Its bifunctional bromide groups make it ideal for industrial chemical synthesis and laboratory-scale organic transformations.
In industrial applications, similar halogenated compounds like 1,2-Dibromoethane, 1,6-Dibromohexane, and 1,3-Dibromopropane are also used for organic synthesis, polymer intermediates, and specialty chemical manufacturing.
Applications of 1,4-Dibromobutane
Pharmaceutical & Fine Chemicals
-
Used as an intermediate in API synthesis and medicinal chemistry
-
Supports the construction of heterocyclic rings and cyclic compounds
-
Applied in the production of bioactive molecules and specialty reagents
Polymer & Material Chemistry
-
Acts as a cross-linking agent in polymer synthesis
-
Used in polyurethane, epoxy, and resin modifications
-
Supports development of high-performance polymeric materials
Organic Synthesis & Specialty Chemicals
-
Serves as a reagent for nucleophilic substitution and cyclization reactions
-
Useful in preparing brominated intermediates for industrial applications
-
Applied in research labs for fine chemical synthesis and experimental reactions
Laboratory & Research Applications
-
Utilized in academic and industrial research labs
-
Supports organic synthesis studies and chemical development projects
-
Applied in synthesis of heterocyclic and functionalized compounds
Safety & Handling Guidelines
-
Avoid contact with skin, eyes, and clothing
-
Use gloves, goggles, and protective clothing
-
Handle in well-ventilated areas or fume hoods
-
Store in tightly sealed containers away from heat, moisture, and strong oxidizers
-
Follow COA and MSDS instructions carefully
Where to Buy 1,4-Dibromobutane?
1,4-Dibromobutane Manufacturer
ChemicalBull supplies high-purity 1,4-Dibromobutane suitable for organic synthesis, polymer chemistry, and specialty chemical production.
1,4-Dibromobutane Supplier & Distributor
-
Available in bulk and laboratory-scale packaging
-
Ideal for industrial chemical synthesis, polymer cross-linking, and fine chemical intermediates
-
COA, MSDS, and detailed technical specifications provided
1,4-Dibromobutane MSDS
-
May cause irritation to skin, eyes, and respiratory system
-
Flammable—keep away from heat, sparks, and open flames
-
Avoid inhalation of vapors and prolonged exposure
-
Store in cool, dry, ventilated conditions
-
Refer to full MSDS for spill control and emergency response procedures
Frequently Asked Questions (FAQs)
-
What is the molecular formula of 1,4-Dibromobutane?
Molecular Formula: C₄H₈Br₂
Molecular Weight: 187.91 g/mol -
What is 1,4-Dibromobutane used for?
Used as a building block in organic synthesis, polymer cross-linking, heterocyclic compound synthesis, and specialty chemical intermediates. -
Which industries use 1,4-Dibromobutane?
Common industries include chemical manufacturing, polymer production, pharmaceuticals, and research laboratories. -
Is 1,4-Dibromobutane hazardous?
Yes. It is flammable and irritant. Proper PPE and safety precautions must be followed. -
Why is it important in organic synthesis?
Its bifunctional bromide groups make it ideal for cyclization, cross-linking, and nucleophilic substitution reactions in industrial and laboratory applications.