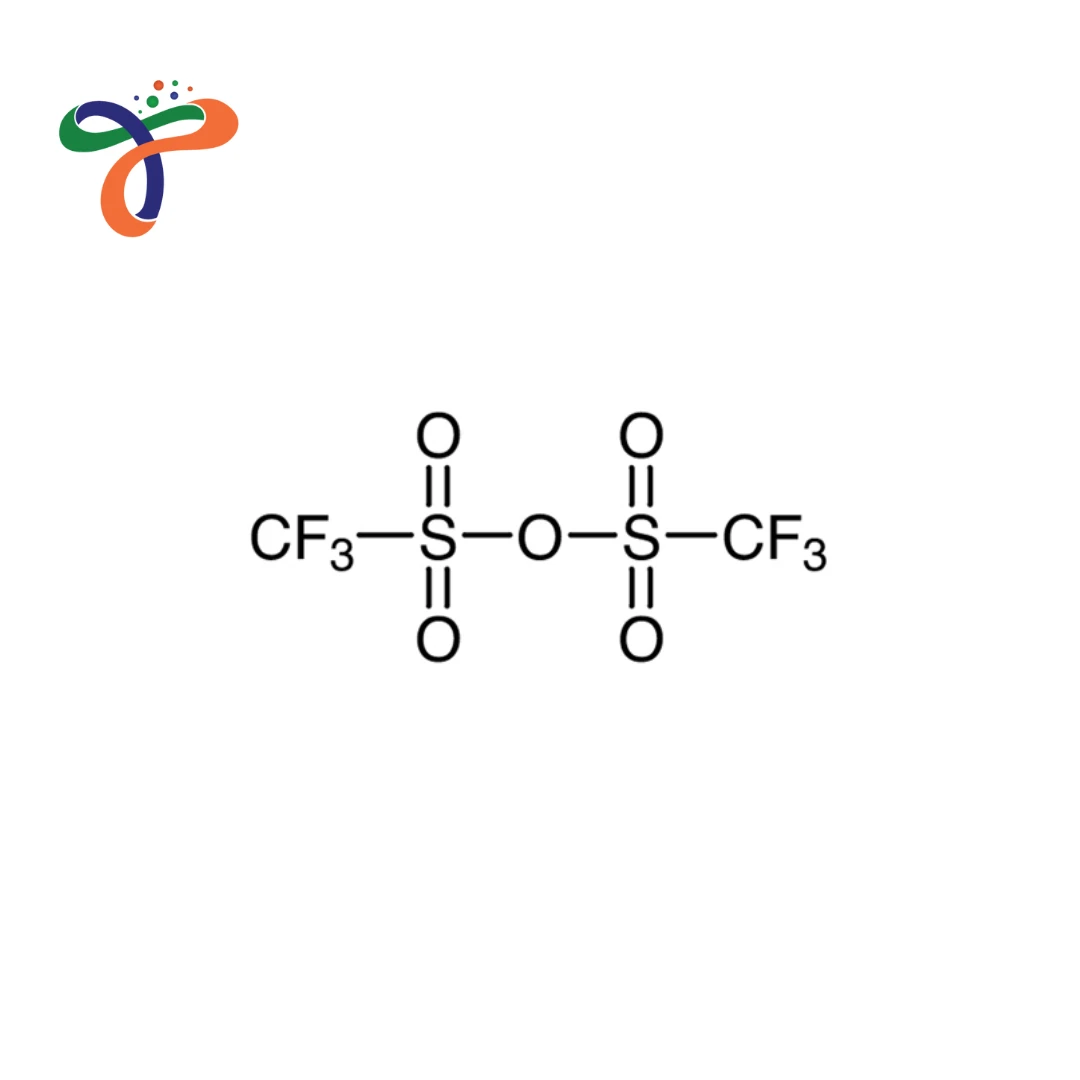
Trifluoromethanesulfonic Anhydride | 358-23-6
Cas No :
358-23-6
Form :
Liquid
Molecular Weight :
282.14 G/Mol
Molecular Formula :
C2F6O5S2
Boiling Point :
82 °C
Melting Point :
-60 °C
Flash Point :
80 °C
Solubility :
Reactive With Water; Soluble In Organic Solvents Such As Dichloromethane And Toluene; Not Soluble In Water.
Description :
Overview of Trifluoromethanesulfonic Anhydride
Trifluoromethanesulfonic Anhydride (CAS: 358-23-6), commonly known as Triflic Anhydride, is a powerful sulfonating and acylating reagent used in organic synthesis, pharmaceutical intermediates, and specialty chemical manufacturing. It appears as a colorless to pale yellow liquid with high reactivity and strong electrophilic properties, making it suitable for activating alcohols, amines, and carboxylic acids.
Trifluoromethanesulfonic Anhydride is widely applied in triflation reactions, esterification, and the preparation of triflate salts. Its high purity ensures reproducible reactions and efficient chemical transformations in both laboratory and industrial settings.
Related products available on ChemicalBull: Trifluoromethanesulfonic Acid, Trifluoromethylsulfonyl Chloride, Methanesulfonyl Chloride – used in pharmaceutical intermediates, sulfonation, and fine chemical synthesis.
Applications of Trifluoromethanesulfonic Anhydride
Organic Synthesis & Triflation Reactions
-
Used to convert alcohols, amines, and phenols into triflate derivatives
-
Facilitates highly selective and efficient chemical transformations
-
Applied in the preparation of pharmaceutical intermediates and specialty chemicals
Pharmaceutical & Fine Chemical Production
-
Key reagent in the synthesis of active pharmaceutical ingredients (APIs)
-
Used in drug discovery and medicinal chemistry for functional group activation
-
Supports multi-step organic reactions and advanced chemical synthesis
Industrial & Laboratory Applications
-
Employed in acylation and sulfonation reactions in chemical industries
-
Used in research laboratories for organic transformations
-
Supports chemical R&D for novel functionalized molecules
Specialty Chemical Applications
-
Helps produce triflate salts and functional intermediates
-
Applied in polymer and advanced material synthesis
-
Supports the development of high-performance chemical derivatives
Safety & Handling Guidelines
-
Highly reactive and corrosive—avoid contact with skin, eyes, and clothing
-
Wear gloves, goggles, and chemical-resistant protective clothing
-
Handle under dry, inert, or ventilated conditions (fume hood recommended)
-
Store in tightly sealed containers away from moisture, bases, and strong oxidizers
-
Follow COA and MSDS instructions strictly
Where to Buy Trifluoromethanesulfonic Anhydride?
Trifluoromethanesulfonic Anhydride Manufacturer
ChemicalBull supplies high-purity Trifluoromethanesulfonic Anhydride for pharmaceutical, organic synthesis, and specialty chemical applications.
Trifluoromethanesulfonic Anhydride Supplier & Distributor
-
Available in laboratory and bulk packaging
-
Ideal for triflation, acylation, and sulfonation reactions
-
COA, MSDS, and detailed product specifications provided
Trifluoromethanesulfonic Anhydride MSDS
-
Causes severe irritation and burns; use PPE
-
Avoid inhalation of vapors and dust
-
Handle under inert atmosphere to prevent reaction with water
-
Store in cool, dry, sealed containers away from incompatible substances
-
Refer to full MSDS for detailed handling, spill control, and emergency procedures
Frequently Asked Questions (FAQs)
-
What is the molecular formula of Trifluoromethanesulfonic Anhydride?
Molecular Formula: C₂F₆O₃S₂
Molecular Weight: 282.13 g/mol -
What is Trifluoromethanesulfonic Anhydride used for?
Used in organic synthesis, triflation reactions, acylation, sulfonation, and pharmaceutical intermediate production. -
Is Trifluoromethanesulfonic Anhydride hazardous?
Yes, highly reactive and corrosive—handle with proper PPE and follow MSDS guidelines strictly.