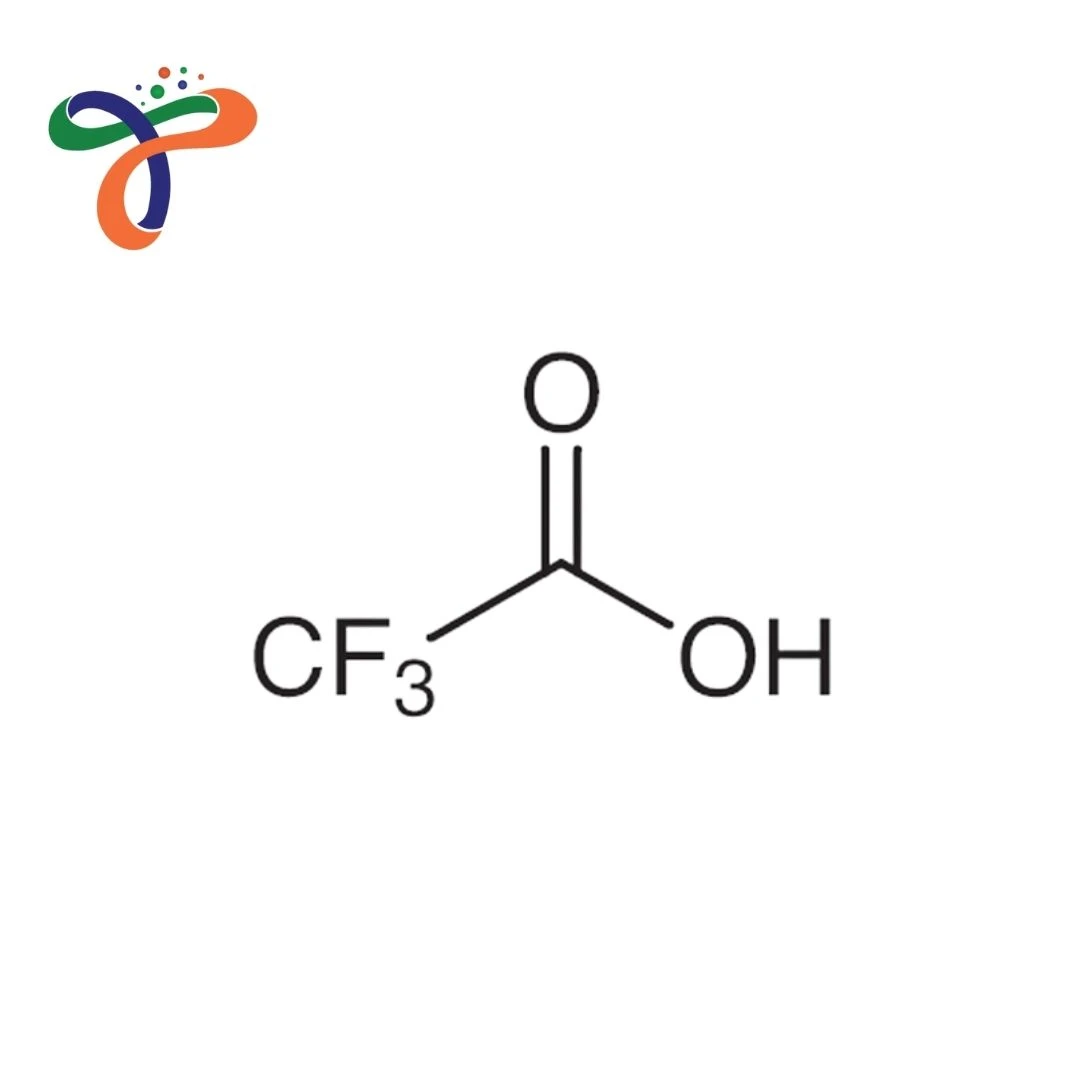
Trifluoroacetic Acid | 76-05-1
Cas No :
76-05-1
Form :
Liquid
Molecular Weight :
114.02
Molecular Formula :
C2HF3O2
Boiling Point :
72,4 °C
Melting Point :
-15.9 °C
Flash Point :
39.0 °C
Solubility :
Miscible With Water; Soluble In Organic Solvents
Description :
Overview of Trifluoroacetic Acid
Trifluoroacetic Acid (TFA) is a strong organic acid used extensively as a reagent in organic synthesis, pharmaceutical manufacturing, and peptide chemistry. Known for its strong acidity (pKa ~0.23), excellent solvating properties, and volatility enabling easy removal, Trifluoroacetic Acid is widely used in cleaving protecting groups, dissolving polymers, and as a solvent and catalyst in various chemical transformations.
This compound appears as a colorless liquid with a pungent, irritating odor. Its three fluorine atoms adjacent to the carboxylic acid make it one of the strongest organic acids, providing unique reactivity in synthesis.
Related products include Trifluoroacetic Anhydride, Acetic Acid, Diethyl Oxalate, and other organic acids and reagents used in pharmaceutical synthesis.
Applications of Trifluoroacetic Acid
Pharmaceutical & Peptide Chemistry
- Used in removing protecting groups (Boc, trityl, etc.)
- Applied in peptide synthesis and deprotection
- Used in cleaving peptides from solid-phase resins
- Applied in pharmaceutical intermediate synthesis
Industrial Chemical Synthesis
- Used as a catalyst in esterification reactions
- Applied in acylation and alkylation reactions
- Used in polymer chemistry and modifications
- Applied in the manufacturing of specialty chemicals
Analytical Chemistry
- Used as a mobile phase additive in HPLC
- Applied in ion-pairing chromatography
- Used in mass spectrometry sample preparation
- Applied in protein and peptide analysis
Safety & Handling Guidelines
- HIGHLY CORROSIVE—causes severe burns
- TOXIC—avoid inhalation of vapors
- Use only in a well-ventilated fume hood
- Wear appropriate PPE, including acid-resistant gloves and a face shield
- Store in tightly closed containers in corrosion-resistant cabinets
- Keep away from bases, metals, and reactive materials
- Have an emergency eyewash and shower immediately available
- Dispose of according to corrosive acid waste protocols
- Follow COA and MSDS instructions strictly
Where to Buy Trifluoroacetic Acid?
Trifluoroacetic Acid Manufacturer
ChemicalBull supplies the highest purity Trifluoroacetic Acid that meets the quality requirements of research and pharmaceuticals.
Trifluoroacetic Acid Supplier & Distributor
- Large quantities of HTML0 are available to be used in pharmaceuticals as well as industrial usage.
- High-purity grades (99%, 99.5%, peptide synthesis grade)
- Special corrosion-resistant packaging
- COA MSDS, as well as full security documentation, are included.
MSDS
With comprehensive warnings about corrosivity as well as toxicity data, along with the strictest handling procedures.
Frequently Asked Questions (FAQs)
-
What is Trifluoroacetic Acid CAS No?
CAS number: 76-05-1. -
What is the structure of Trifluoroacetic Acid?
CF₃COOH; carboxylic acid with three fluorine atoms on the adjacent carbon; molecular formula C₂HF₃O₂. -
What is the Trifluoroacetic Acid formula?
Molecular formula: C₂HF₃O₂ or CF₃COOH with molecular weight 114.02 g/mol.