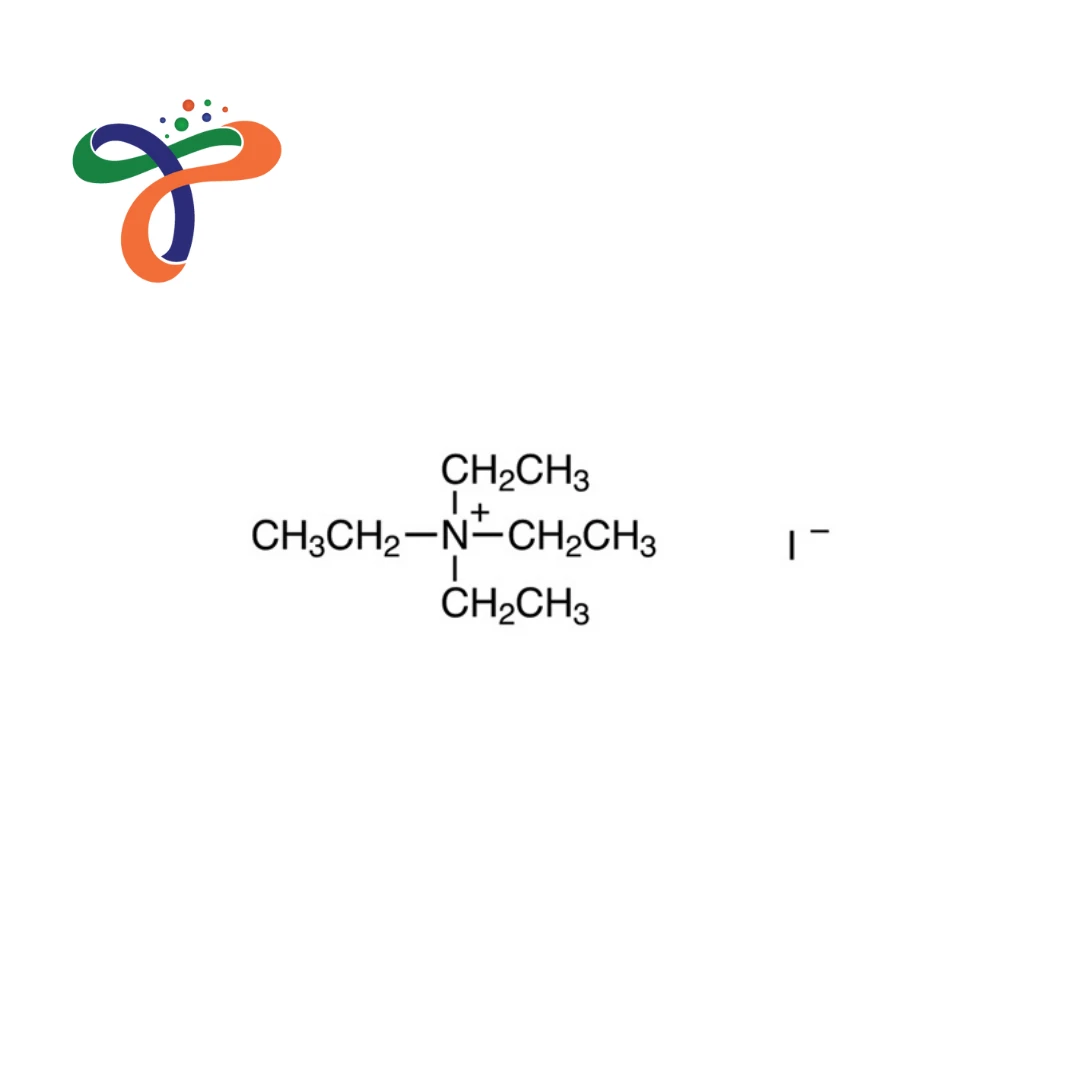
Tetraethylammonium Iodide | 68-05-3
Cas No :
68-05-3
Form :
Solid
Molecular Weight :
257.16 G/Mol
Molecular Formula :
C8H20IN
Boiling Point :
300 °C
Melting Point :
190 °C
Flash Point :
150 °C
Solubility :
Soluble In Water; Soluble In Methanol; Soluble In Ethanol; Insoluble In Nonpolar Solvents
Description :
Overview of Tetraethylammonium Iodide
Tetraethylammonium Iodide (CAS: 71-55-6) is a quaternary ammonium salt widely used in phase transfer catalysis, organic synthesis, electrochemistry, and specialty chemical research. It appears as a white crystalline solid or powder and is valued for its high solubility in polar solvents and ionic conductivity, making it a versatile reagent in laboratory and industrial applications.
Tetraethylammonium Iodide is primarily employed in organic transformations, halide exchange reactions, and as a supporting electrolyte in electrochemical studies. Its quaternary ammonium structure allows it to act as a phase transfer catalyst, enhancing reaction rates between immiscible phases. Related compounds such as Tetraethylammonium Bromide, Tetraethylammonium Chloride, and Quaternary Ammonium Salts are also used in organic synthesis, electrochemical applications, and specialty chemical formulations.
Applications of Tetraethylammonium Iodide
Organic Synthesis
-
Used as a phase transfer catalyst for nucleophilic substitution and elimination reactions
-
Facilitates halide exchange and quaternization reactions
-
Supports synthesis of fine chemicals and specialty organic intermediates
Electrochemistry & Energy Applications
-
Applied as a supporting electrolyte in electrochemical cells
-
Enhances ionic conductivity in non-aqueous electrochemical systems
-
Used in research involving redox reactions and electrodeposition
Laboratory & Research Applications
-
Widely used in academic and industrial R&D laboratories
-
Supports reaction optimization, catalyst studies, and organic transformations
-
Useful in developing quaternary ammonium-based reagents
Specialty Chemical Manufacturing
-
Employed in preparation of quaternary ammonium salts and intermediates
-
Used in fine chemical synthesis and reagent production
-
Supports pharmaceutical, agrochemical, and polymer research applications
Safety & Handling Guidelines
-
May cause skin, eye, and respiratory irritation
-
Handle in well-ventilated areas
-
Use protective gloves, goggles, and lab coats
-
Store in airtight containers, away from strong oxidizers and acids
-
Avoid exposure to moisture and incompatible chemicals
-
Follow COA and MSDS instructions strictly
Where to Buy Tetraethylammonium Iodide?
Tetraethylammonium Iodide Manufacturer
ChemicalBull supplies high-purity Tetraethylammonium Iodide suitable for organic synthesis, electrochemistry, and phase transfer catalysis.
Tetraethylammonium Iodide Supplier & Distributor
-
Available in laboratory and bulk packaging
-
Ideal for organic reactions, quaternary ammonium studies, and specialty chemical synthesis
-
COA, MSDS, and technical specifications provided
Tetraethylammonium Iodide MSDS
-
Causes mild to moderate irritation to skin, eyes, and respiratory tract
-
Handle with proper PPE and ventilation
-
Store in cool, dry areas away from incompatible chemicals
-
Refer to full MSDS for spill control and emergency procedures
Frequently Asked Questions (FAQs)
-
What is the molecular formula of Tetraethylammonium Iodide?
Molecular Formula: C₈H₂₀IN
Molecular Weight: 228.17 g/mol -
What is Tetraethylammonium Iodide used for?
Used primarily in phase transfer catalysis, organic synthesis, electrochemical studies, and specialty chemical research. -
Is Tetraethylammonium Iodide hazardous?
Yes. It may cause skin, eye, and respiratory irritation. Proper PPE and safety precautions are recommended. -
Which industries commonly use Tetraethylammonium Iodide?
Used in academic and industrial research laboratories, fine chemical manufacturing, electrochemical studies, and specialty chemical synthesis. -
Why is it important in organic synthesis?
It enhances reaction rates in biphasic systems and facilitates halide exchange, quaternization, and other organic transformations.