Sodium Peroxide | 1313-60-6
Cas No :
1313-60-6
Form :
Powder
Molecular Weight :
78.0 G/Mol
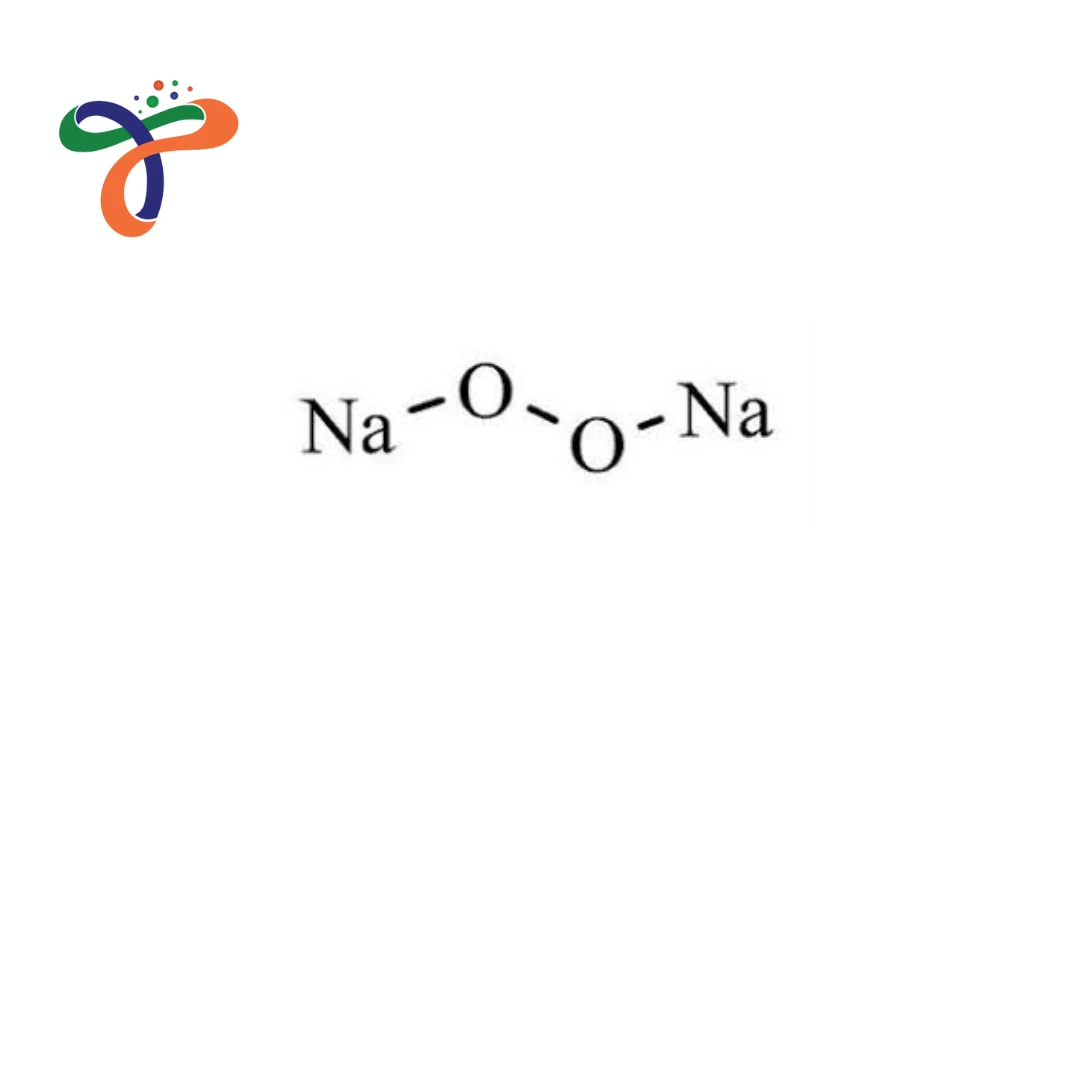
Molecular Formula :
NA2O2
Boiling Point :
500 °C
Melting Point :
470 °C
Flash Point :
300 °C
Solubility :
Soluble In Water; Reacts With Water To Release Oxygen
Description :
Overview of Sodium Peroxide
Sodium Peroxide can be described as a powerful inorganic oxidizing chemical widely employed in chemical synthesis, bleaching as well as environmental treatment and the oxidation process in laboratories. It is typically available in a light yellow solid that reacts strongly in water, releasing oxygen and create sodium hydroxide.
As an industrial chemical, Sodium Peroxide is valued for its powerful oxidizing properties and ability to generate oxygen in controlled systems. It is used in regulated industrial environments where strong oxidation capability is required.
Applications of Sodium Peroxide
Chemical Oxidation Processes
Sodium Peroxide is extensively used as an oxidizing agent in organic and inorganic chemical synthesis. It supports oxidation reactions and intermediate production in industrial chemical plants.
It may be processed alongside Hydrogen Peroxide, Sodium Hydroxide Prills, and Sulfuric Acid under controlled reaction conditions.
Bleaching & Pulp Treatment
The chemical is utilized in bleaching processes, especially in specialty textile and pulp processing where controlled oxidation increases the whiteness of the material and improves its quality.
It supports industrial-scale bleaching systems.
Environmental & Waste Treatment
Sodium Peroxide is applied in environmental treatment processes for oxidation of contaminants and hazardous waste neutralization.
It contributes to industrial effluent management systems requiring strong oxidizing performance.
Oxygen Generation Systems
Due to its ability to release oxygen upon reaction with water or carbon dioxide, Sodium Peroxide is used in controlled oxygen-generating systems in specialized industrial applications.
Safety & Handling Guidelines
- Store in a cool, dry, and moisture-free environment
- Keep containers tightly sealed and protected from humidity
- Avoid contact with water unless under controlled conditions
- Prevent contact with organic materials
- Use protective gloves, goggles, and appropriate industrial clothing
- Handle under strict industrial safety protocols
- Dispose of waste according to hazardous material regulations
Sodium Peroxide is a strong oxidizer and reacts vigorously with water. Always consult the MSDS before industrial handling.
Where to Buy Sodium Peroxide?
Sodium Peroxide Manufacturer
Sodium Peroxide is manufactured under controlled industrial standards to ensure consistent purity and oxidizing performance for chemical and environmental industries.
Sodium Peroxide Supplier & Distributor
Sodium Peroxide is available in bulk industrial packaging formats. Buy Sodium Peroxide from Chemicalbull Pvt. Ltd., a trusted industrial chemicals supplier and distributor in India, ensuring reliable sourcing and compliant logistics support.
MSDS for Sodium Peroxide
The MSDS for Sodium Peroxide provides detailed information on hazard identification, safe handling procedures, storage requirements, exposure controls, and emergency response measures. It must be reviewed prior to use.
Frequently Asked Questions (FAQs)
-
What is Sodium Peroxide used for?
Sodium Peroxide is used as an oxidizing agent in chemical synthesis, bleaching processes, environmental treatment, and oxygen generation systems. -
What is another name for Sodium Peroxide?
It is sometimes referred to as disodium peroxide in technical terminology. -
What are the side effects of Sodium Peroxide?
Exposure may cause severe irritation or burns to skin, eyes, and respiratory tract due to its strong oxidizing and alkaline nature. Proper industrial protective measures are essential. -
Is Sodium Peroxide soluble in water?
It reacts with water rather than simply dissolving, forming sodium hydroxide and releasing oxygen. -
What is the formula and structure of Sodium Peroxide?
The chemical formula is Na₂O₂, and it consists of sodium cations (Na⁺) and a peroxide anion (O₂²⁻) with an oxygen–oxygen single bond in its ionic lattice structure.