Pyrrole | 109-97-7
Cas No :
109-97-7
Synonyms :
Form :
Liquid
Molecular Weight :
67.09 G/Mol
Molecular Formula :
C4H5N
Boiling Point :
128 °C
Melting Point :
-60 °C
Flash Point :
-20 °C
Solubility :
Soluble In Water And Many Organic Solvents
Description :
Overview of Pyrrole
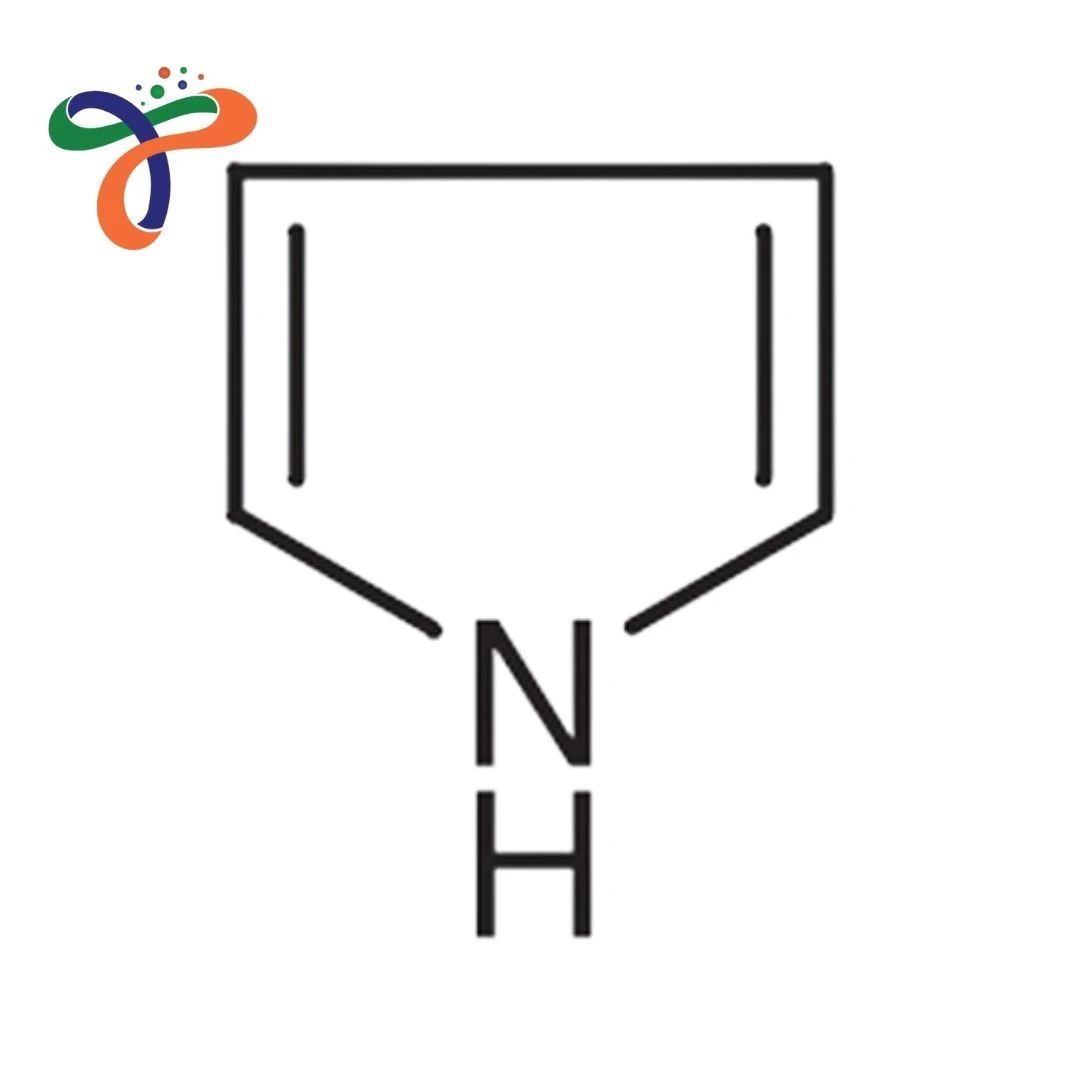
Pyrrole is a colorless to yellowish volatile liquid heterocyclic aromatic compound that darkens on exposure to air, forming the core of many natural products and pharmaceuticals.
Chemical Formula: C4H5N Pyrrole formula: C4H4NH (often written as such) Pyrrole structure: Five-membered ring with four carbons and one nitrogen, aromatic with 6 π-electrons Functional properties: Weakly basic and acidic, highly reactive toward electrophilic substitution at α-positions
Application of Pyrrole
Organic Synthesis as well as Materials
- What is Pyrrole used for? The building block used to make porphyrins, chlorophyll-like analogs, conductor polymers (polypyrrole), and pharmaceuticals.
- Pyrrole can be used as a solvent in Agrochemicals, dyes, and as an intermediate.
Industrial Use
- It is used to synthesize fine chemicals and heterocyclic derivatives
- Sometimes used in reaction with esters, such as Diethyl Maleate, to make Diels-Alder, or in condensate products
Safe Handling and Safety Guidelines
- Use protective glasses, gloves, and other appropriate attire
- Avoid contact with the skin or breathing (volatile as well as toxic)
- Make use of it only in an area that is well ventilated, such as a fume hood
- Store in sealed, cool, dark containers to prevent darkening/resinification
- Use MSDS guidelines and safety guidelines
Where can I buy Pyrrole?
Pyrrole Manufacturer
- High-purity Pyrrole Suppliers for industrial and research synthesizing
Pyrrole Supplier & Distributor
- Bulk supply complete with COA, TDS, MSDS, and COA. MSDS available
Pyrrole MSDS
Offers information on dangers, handling, storage, as well as emergency measures.
Frequently Asked Questions (FAQs)
-
What is the Pyrrole formula and structure?
C4H5N; five-membered aromatic heterocycle with nitrogen and conjugated double bonds. -
What are the uses of Pyrrole?
As a precursor to polymers, pharmaceuticals, dyes, and natural product analogs, such as heme/chlorophyll. -
What is Pyrrole used for in synthesis?
Key intermediates in heterocyclic chemistry, porphyrins, and conducting materials.