Potassium Sulfide | 1312-73-8
Cas No :
1312-73-8
Form :
Solid
Molecular Weight :
110.26 G/Mol
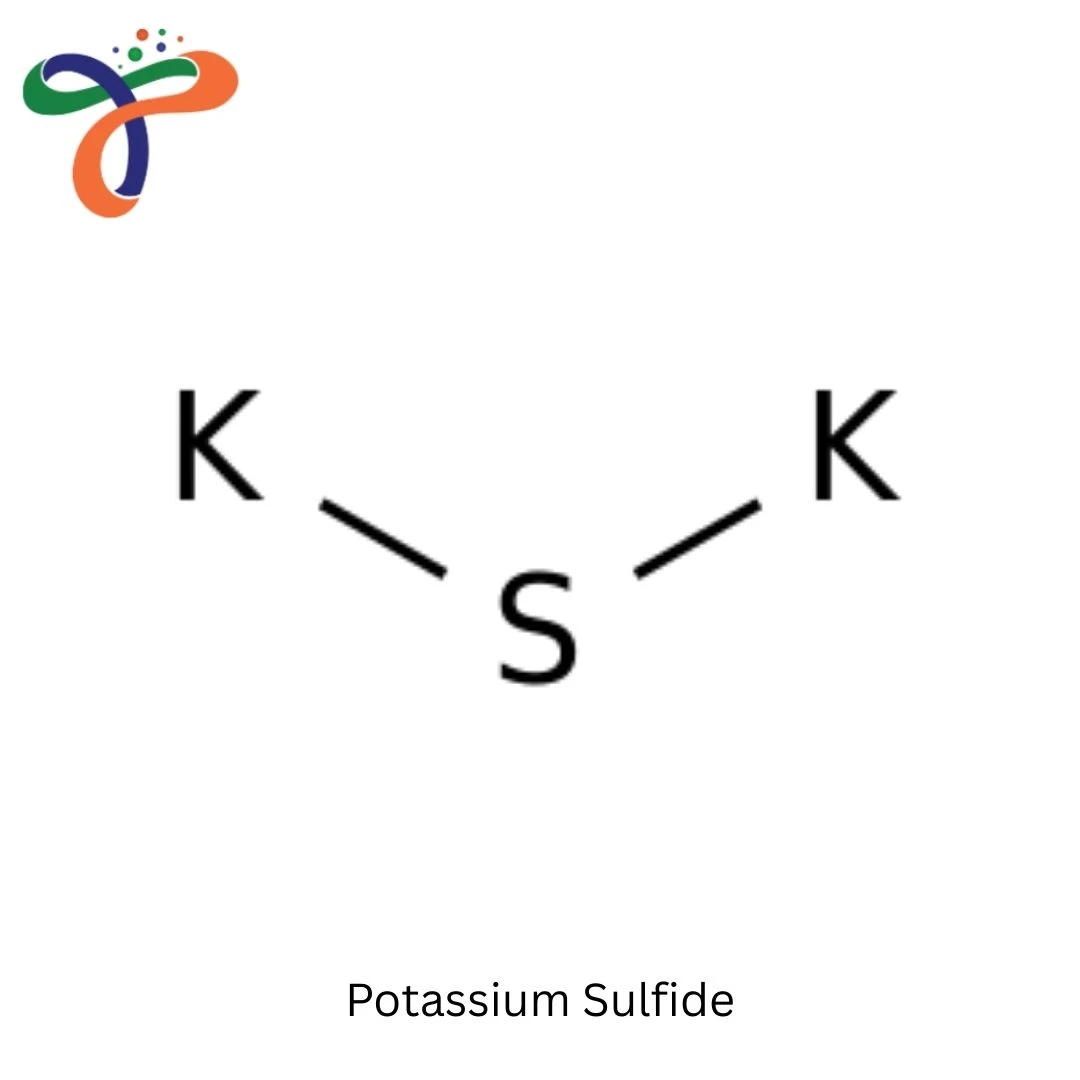
Molecular Formula :
K2S
Boiling Point :
1100 °C
Melting Point :
900 °C
Flash Point :
9999 °C
Solubility :
Soluble In Water; Hydrolyzes In Water To Potassium Hydroxide And Hydrogen Sulfide
Description :
Overview of Potassium Sulfide
Potassium Sulfide (CAS: 1312-73-8) is an important chemical compound widely used in chemical synthesis, water treatment, and industrial applications. It appears as a pale yellow crystalline solid and is known for its strong reducing properties and ability to react with moisture to release hydrogen sulfide gas. Potassium Sulfide is primarily used in the production of potassium hydrosulfide, polysulfides, and other sulfur-based chemicals.
This compound is highly valued in mineral extraction, fertilizer production, and chemical manufacturing, making it essential in various industrial processes. Its reactivity and versatility also make it useful for water purification, leather tanning, and dyeing applications. Potassium Sulfide is also found in products like Calcium Sulfide, Sodium Sulfide, and Lithium Sulfide. These compounds share similar properties and applications in various chemical industries.
Applications of Potassium Sulfide
Chemical Manufacturing & Synthesis
-
Used in the production of potassium hydrosulfide and polysulfides
-
Serves as a reducing agent in organic synthesis and polymer production
-
Supports the production of sulfur-based compounds, including sulfonates and thiols
Water Treatment & Environmental Applications
-
Applied in water purification and heavy metal removal
-
Used to precipitate metal ions from wastewater, improving water quality
-
Essential in wastewater treatment systems to remove sulfur contaminants
Fertilizer Production
-
Used in potassium-based fertilizers for supplying essential potassium and sulfur nutrients
-
Helps in soil conditioning and improving plant growth
-
Widely used in the agricultural sector for sulfur-enhanced fertilizers
Leather & Textile Industries
-
Used in leather tanning to enhance flexibility and durability of leather products
-
Applied in the dyeing process to improve color fixation and prevent discoloration
-
Enhances the quality and longevity of textiles in the manufacturing process
Specialty Chemical Manufacturing
-
Utilized in the production of specialty sulfur compounds and fine chemicals
-
Supports organic synthesis by acting as a nucleophilic reagent
-
Plays a key role in custom chemical formulations for industrial and laboratory use
Safety & Handling Guidelines
-
Highly reactive – handle in dry, moisture-free environments
-
Wear protective gloves, goggles, and appropriate PPE when handling
-
Store in airtight containers and away from moisture, heat, and oxidizing agents
-
Use in well-ventilated areas to avoid hydrogen sulfide exposure
Potassium Sulfide MSDS
-
Toxic and can release hydrogen sulfide gas upon contact with moisture
-
May cause skin and eye irritation and respiratory distress if inhaled
-
Handle with proper PPE and store in cool, dry conditions
-
Refer to full MSDS for emergency response and spill management
Where to Buy Potassium Sulfide?
Potassium Sulfide Manufacturer
ChemicalBull supplies high-purity Potassium Sulfide suitable for chemical manufacturing, water treatment, and industrial applications.
Potassium Sulfide Supplier & Distributor
-
Available in bulk and laboratory-scale packaging
-
Ideal for fertilizer production, water treatment, and chemical synthesis
-
COA, MSDS, and detailed technical specifications provided
Frequently Asked Questions (FAQs)
-
What is the molecular formula of Potassium Sulfide?
Molecular Formula: K₂S
Molecular Weight: 94.19 g/mol -
What is Potassium Sulfide used for?
Potassium Sulfide is used in chemical synthesis, water treatment, fertilizer production, and leather tanning. -
Is Potassium Sulfide hazardous?
Yes, it is toxic and should be handled carefully with proper PPE. It can release hydrogen sulfide gas in the presence of moisture. -
Which industries use Potassium Sulfide?
Industries involved in chemical manufacturing, agriculture, textile, leather, and environmental remediation commonly use Potassium Sulfide. -
How should Potassium Sulfide be stored?
Store in airtight containers in cool, dry conditions away from moisture and oxidizing agents.