Phenyl Chloroformate | 1885-14-9
Cas No :
1885-14-9
Synonyms :
Form :
Liquid
Molecular Weight :
156.57
Molecular Formula :
C7H5CLO2
Boiling Point :
74 - 75 °C
Melting Point :
25 °C
Flash Point :
69 °C
Description :
Overview of Phenyl Chloroformate
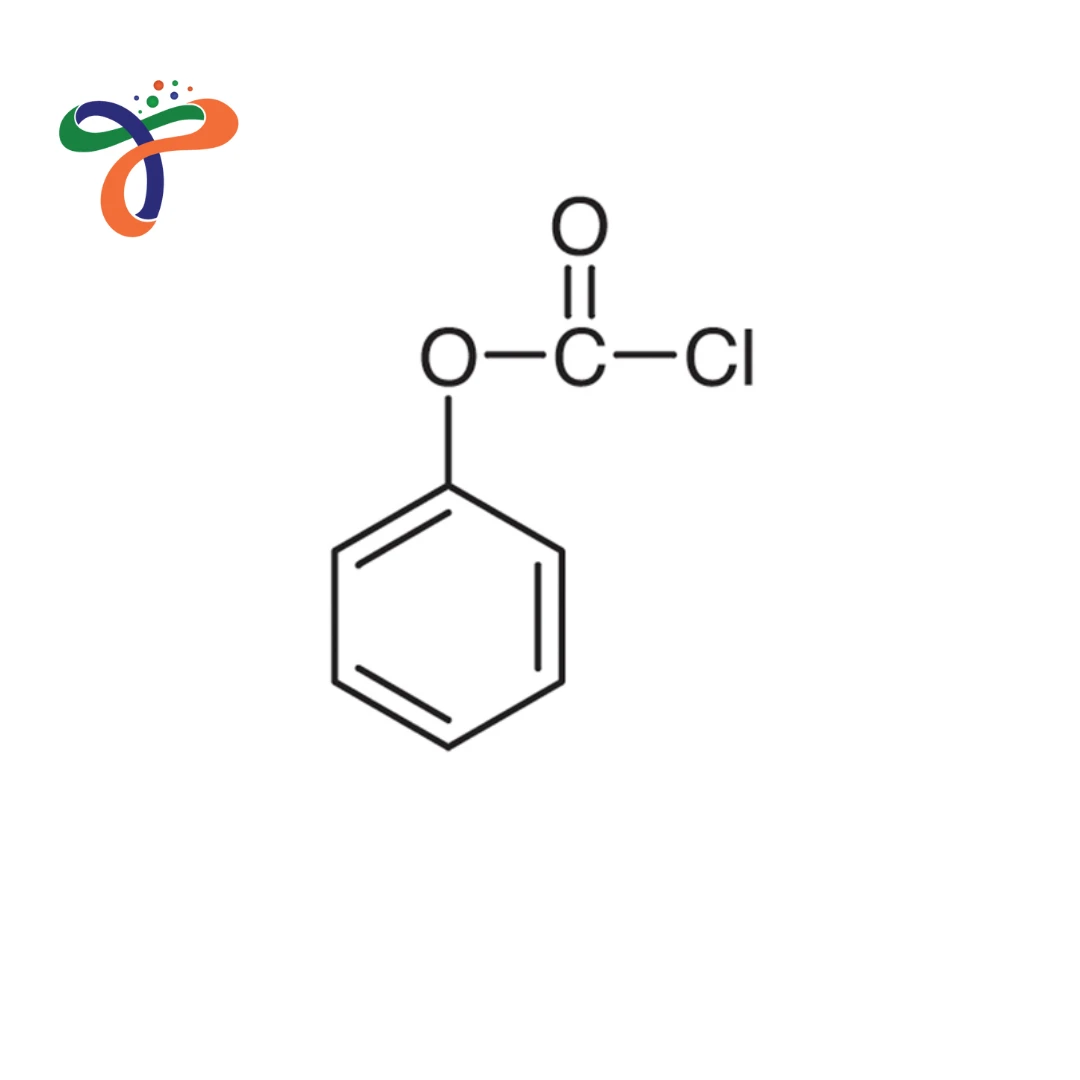
Phenyl Chloroformate (also known as phenyl chlorocarbonate) is a colourless to pale yellow liquid chloroformate ester, a reactive acylating agent used in protection and derivatisation.
Chemical Formula: C7H5ClO2 or ClCOOC6H5 Molecular weight: 156.57 g/mol
CAS No: 1885-14-9 Functional properties: Highly reactive with nucleophiles, releases HCl, used for carbamate formation
Applications of Phenyl Chloroformate
Organic Synthesis
-
Protects compounds such as phenyl carbamates and for the synthesis of carbonates as well as Urethanes
-
Precursor to polycarbonates and polyurethanes, as well as pharmaceutical intermediates
Industrial Use
-
For the production of speciality polymers as well as fine chemicals
-
Related to epoxide or silane modifications in coatings involving Cyclohexene Oxide or Dodecyltrimethoxysilane
Safety and Handling Guidelines
-
Use protective glasses, gloves, and wear appropriate clothes (corrosive or poisonous smokes )
-
Avoid contact with the skin for a prolonged period, as well as inhalation
-
Only use inside a ventilated fume hood
-
Storage in sealed containers free of bases and moisture
-
Use MSDS guidelines as well as standard safety guidelines (lachrymatory toxic, lachrymatory)
Where to Buy Phenyl Chloroformate
Phenyl Chloroformate Manufacturer
-
High-quality suppliers for synthesis and industrial grades
Phenyl Chloroformate Supplier & Distributor
-
Bulk supply with COA, TDS & MSDS available
Phenyl Chloroformate MSDS
Provides details on hazards, handling, storage, and emergency precautions
Frequently Asked Questions (FAQs)
-
What is the Phenyl chloroformate formula and CAS?
ClCOOC6H5 (C7H5ClO2), molecular weight 156.57 g/mol, CAS 1885-14-9. -
What are the industrial uses of Phenyl chloroformate?
It is used as an acylating/protecting agent in carbamates, carbonates, polymers, and pharmaceutical synthesis. -
How is Phenyl chloroformate handled safely?
Use PPE, work in a fume hood, and avoid moisture (hydrolyses to phenol and HCl).