N-Butyl Bromide | 109-65-9
Cas No :
109-65-9
Synonyms :
Form :
Liquid
Molecular Weight :
137.02 G/Mol
Molecular Formula :
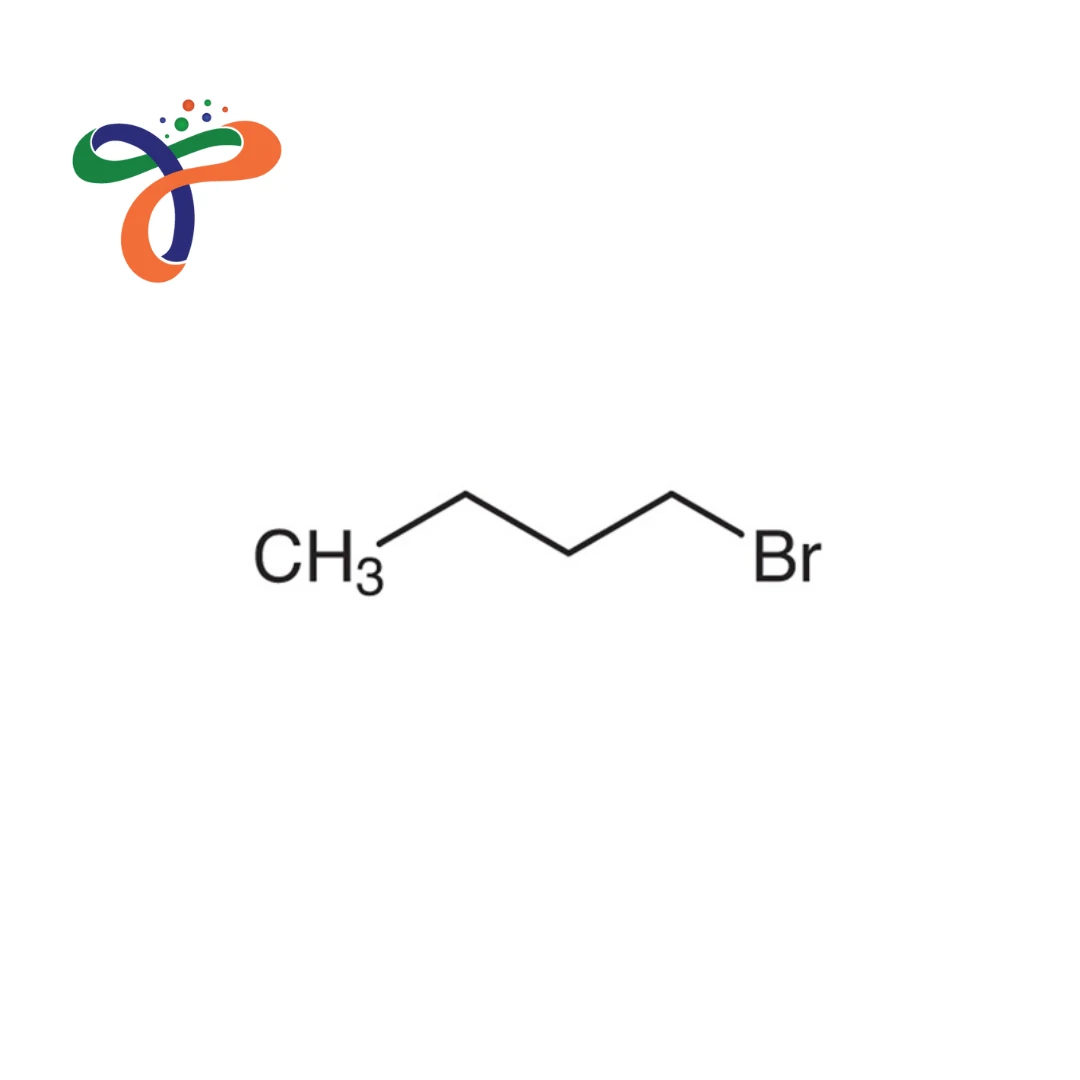
C4H9BR
Description :
Overview of N-Butyl Bromide
N-Butyl Bromide (also called 1-bromobutane) is an organobromine compound. It's a transparent liquid with a yellowish hue that serves to act as an alkylating agent for organic syntheses.
Chemical Formula C4H9Br, butyl bromide structure CH3(CH2)3Br. An alkyl chain that is linear with bromine at the end. Properties of use: Extremely reactive in nucleophilic changes, inflammable
Applications of N-Butyl Bromide
Organic Synthesis
- N-butyl Bromide can be used as an alkylating agent to introduce butyl groups
- Its structure, butyl bromide, facilitates Grignard reactions as well as pharmaceutical intermediates
Industrial Use
- Synthesis of N-Butyl Bromide from butanol and HBr
- Utilised in agrochemicals, pharmaceuticals, and even as an agent for solvent
Safety and Handling Guidelines
- Protect yourself with goggles, gloves, and other appropriate attire
- Avoid prolonged contact with skin as well as inhalation
- Make use of it only in a ventilated area, such as a fume hood
- Keep sealed containers far from sources of heat
- Be sure to follow MSDS and safety guidelines
Where to Buy N-Butyl Bromide
N-Butyl Bromide Manufacturer
- Suppliers of high-purity n-butyl bromide for synthesis
N-Butyl Bromide Supplier & Distributor
- Bulk supply with COA, TDS & MSDS available
N-Butyl Bromide MSDS
Provides details on hazards, handling, storage, and emergency precautions
Frequently Asked Questions (FAQs)
-
What is n-butyl bromide used for?
It is used as an alkylating agent in organic synthesis, pharmaceuticals, and the production of chemical intermediates. -
What is the structure of n-butyl bromide?
Linear chain CH3(CH2)3Br, with bromine attached to the primary carbon. - How is the synthesis of n-butyl bromide performed?
Typically, by reacting n-butanol with hydrobromic acid under controlled conditions.