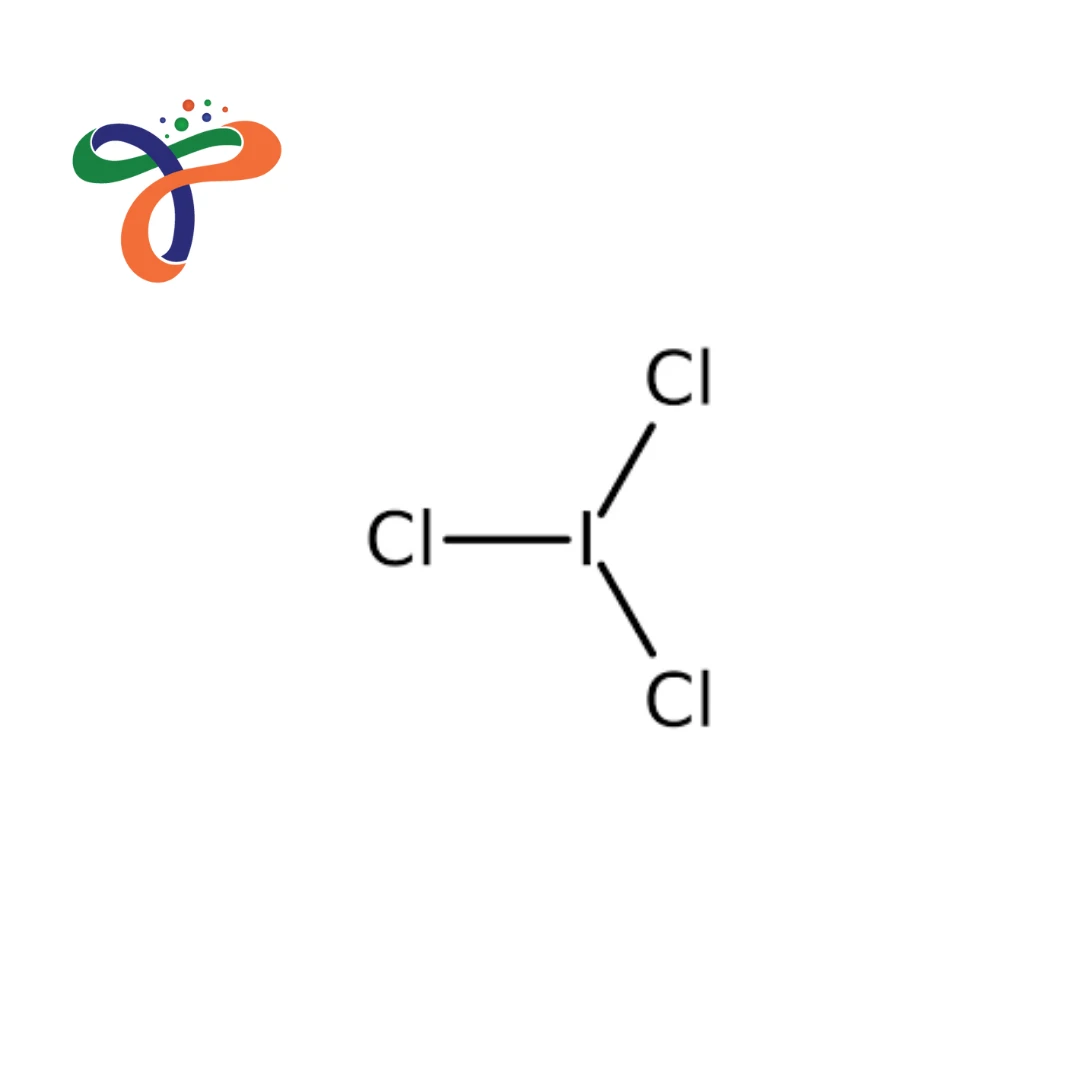
Iodine Trichloride | 865-44-1
Cas No :
865-44-1
Form :
Solid
Molecular Weight :
233.26 G/Mol
Molecular Formula :
ICL3
Boiling Point :
45 °C
Melting Point :
-10 °C
Flash Point :
150 °C
Solubility :
Reactive With Water; Soluble In Nonpolar Solvents Such As Carbon Tetrachloride
Description :
Overview of Iodine Trichloride
Iodine Trichloride (CAS: 7790-98-9) is a highly reactive interhalogen compound widely used in organic synthesis, analytical chemistry, and specialty chemical applications. It appears as a yellow to reddish-brown crystalline solid or liquid with a pungent odor and is valued for its strong oxidizing and chlorinating properties.
Iodine Trichloride is primarily employed in halogenation reactions, organic transformations, and chemical intermediate preparation. Its unique ability to transfer both iodine and chlorine atoms makes it useful for producing iodo-chloro derivatives, pharmaceuticals, dyes, and fine chemicals. Related interhalogen compounds such as Iodine Monochloride (ICl), Iodine Pentafluoride (IF₅), and Chlorine Trifluoride (ClF₃) are also used in specialty organic and inorganic chemical synthesis.
Applications of Iodine Trichloride
Organic & Pharmaceutical Synthesis
-
Used as a halogenating reagent for aromatic and aliphatic compounds
-
Supports synthesis of iodo-chloro derivatives in drug intermediates
-
Applied in production of pharmaceutical intermediates and fine chemicals
Analytical & Laboratory Applications
-
Employed as an oxidizing agent in chemical analysis
-
Used in laboratory experiments requiring selective halogenation
-
Suitable for research and development of novel chemical compounds
Specialty Chemical Manufacturing
-
Applied in dye and pigment intermediate synthesis
-
Useful in fine chemical and specialty chemical formulations
-
Supports production of functionalized halogenated compounds
Industrial Applications
-
Used in chemical industry for selective chlorination and iodination
-
Supports oxidation and halogenation processes
-
Applied in high-value chemical intermediate production
Safety & Handling Guidelines
-
Highly oxidizing and corrosive; avoid contact with skin, eyes, and clothing
-
Handle in well-ventilated areas or fume hoods
-
Use protective gloves, goggles, and lab safety equipment
-
Store in airtight, corrosion-resistant containers away from moisture and heat
-
Avoid contact with reducing agents and combustible materials
-
Follow COA and MSDS instructions strictly
Where to Buy Iodine Trichloride?
Iodine Trichloride Manufacturer
ChemicalBull supplies high-purity Iodine Trichloride suitable for organic synthesis, analytical chemistry, and specialty chemical manufacturing.
Iodine Trichloride Supplier & Distributor
-
Available in laboratory and bulk industrial quantities
-
Ideal for halogenation reactions, pharmaceutical intermediates, and fine chemical synthesis
-
COA, MSDS, and technical specifications provided
Iodine Trichloride MSDS
-
May cause severe irritation to skin, eyes, and respiratory tract
-
Reacts violently with organic and reducing materials
-
Toxic fumes may be released upon decomposition
-
Store in cool, dry, well-ventilated areas away from incompatible substances
-
Refer to full MSDS for emergency response and spill handling
Frequently Asked Questions (FAQs)
-
What is the molecular formula of Iodine Trichloride?
Molecular Formula: ICl₃
Molecular Weight: 232.25 g/mol -
What is Iodine Trichloride used for?
Used primarily as a halogenating and oxidizing agent in organic synthesis, pharmaceuticals, dyes, and specialty chemicals. -
Is Iodine Trichloride hazardous?
Yes, it is highly reactive, corrosive, and oxidizing; proper PPE and handling procedures are required. -
Which industries commonly use Iodine Trichloride?
Pharmaceutical, fine chemical, specialty chemical, and research laboratories frequently use it for halogenation and intermediate production. -
Why is it preferred in halogenation reactions?
It effectively transfers both iodine and chlorine atoms, making it valuable for selective halogenation of organic compounds.