Ethyl Cyanoacetate | 105-56-6
Cas No :
105-56-6
Form :
Liquid
Molecular Weight :
113.11 G/Mol
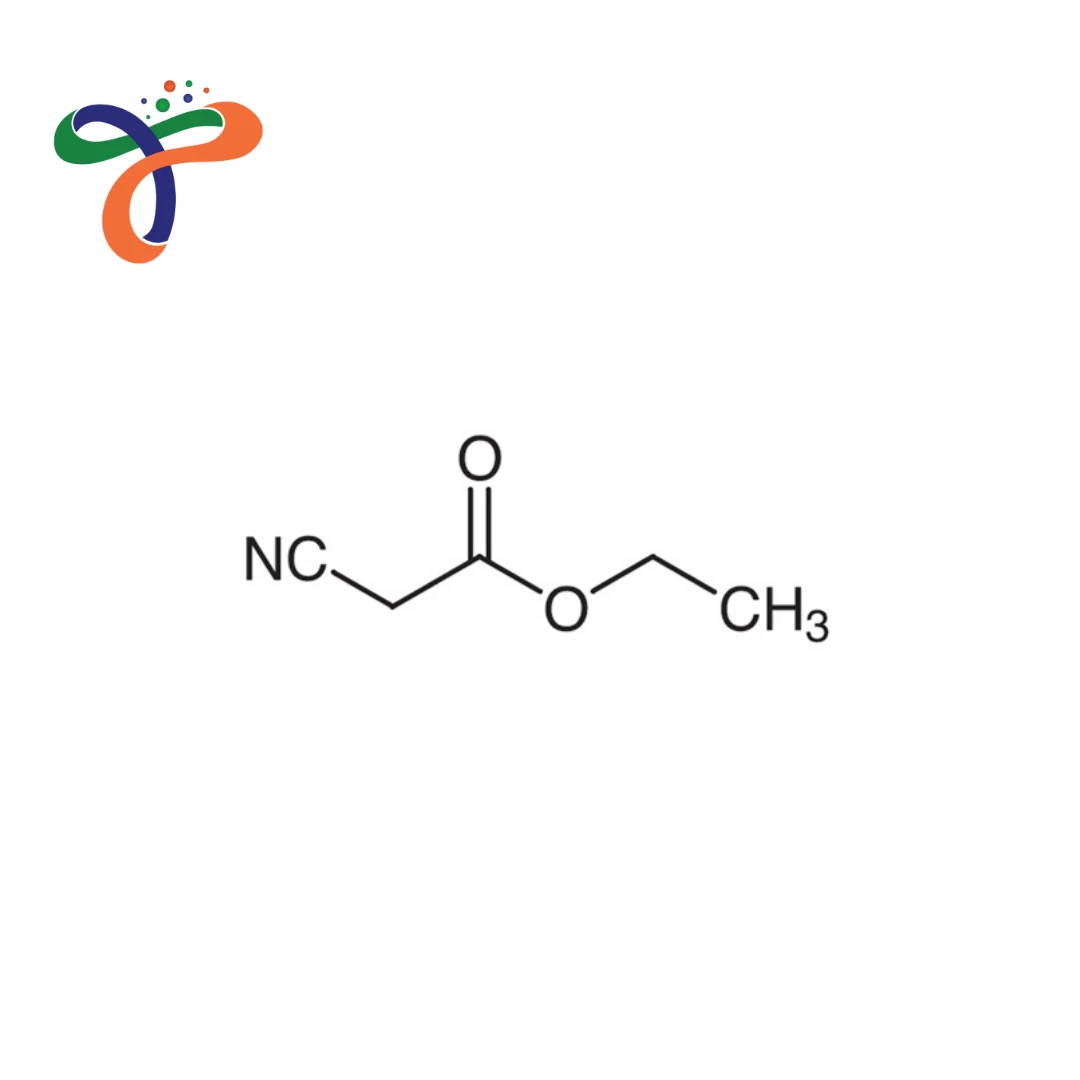
Molecular Formula :
C5H7NO2
Boiling Point :
208 - 210 °C
Melting Point :
22 °C
Flash Point :
110 °C
Solubility :
Chloroform (Slightly), Ethyl Acetate (Very Slightly), Methanol (Slightly)
Description :
Overview of Ethyl Cyanoacetate
Ethyl Cyanoacetate can be described as a multi-functional and versatile compound, used as an intermediate in pharmaceuticals, a chemical building block and an organic methylene reactive compound. The compound is known for its dual functions (nitrile and ester group activating the chemical methylene) as well as its excellent reactivity in the condensation reaction. Ethyl Cyanoacetate is extensively used for the synthesis of pharmaceuticals, agricultural chemical production, and the manufacturing of different organic chemical compounds.
The compound is a transparent, pale yellow to light colour and has a light, pleasant scent. The methylene group that is activated (flanked with both nitrile and ester groups) is extremely reactive in Knoevenagel condensation, Michael addition, and many other important chemical changes.
Other related products include Malononitrile, Diethyl Malonate, Ethyl Acetoacetate and many other methylene-based active compounds utilised in fine chemical and pharmaceutical syntheses.
Applications of Ethyl Cyanoacetate
Pharmaceutical Synthesis
- Utilised in the process of synthesising different pharmaceutical intermediates
- Applied in preparing pyrimidine derivatives for drugs
- It is used in the manufacture of Cephalosporin antibiotic intermediates
- It is used in synthesising cardiovascular medications
- Utilised in the creation of anti-inflammatory chemicals
- Applied in developing CNS active pharmaceuticals
- It is used in the synthesis of the antihypertensive drug intermediates
- It is used in the manufacture of antiparasitic chemicals
Organic Synthesis
- It is used for Knoevenagel condensation reactions
- In Michael's addition reactions
- Utilised for Claisen condensation reactions
- It is used in the Gewald reaction to Thienopyrimidines
- It is used in the preparation of the cyanoacetyl derivatives
- It is used in the process of synthesising heterocyclic compounds
- It is used in the preparation of amino acid analogues
- Utilised in the overall production of bioactive natural substances
Agrochemical and Industrial
- It is used in the synthesis of herbicide intermediates
- Used in the manufacturing of the fungicide compounds
- It is used to make special pigments and dyes
- Used in the preparation of adhesive formulations
- Utilised in the development of the performance chemicals
- Used in the production of intermediates for cyanoacrylate adhesives
Handling and Safety Guidelines
- Beware of inhaling vapours, which can be toxic to inhale
- Wear gloves, goggles and appropriate clothing when handling
- Handle only in ventilated areas or with a fume hood.
- Storage in securely sealed containers, away from light and heat
- Stay clear of powerful bases, acids and agents that oxidise
- Beware of contact with the eyes or skin
- Use the COA as well as the MSDS guidelines completely
Where to Buy Ethyl Cyanoacetate?
Ethyl Cyanoacetate Manufacturer
ChemicalBull supplies high-purity Ethyl Cyanoacetate, meeting pharmaceutical and research quality standards.
Ethyl Cyanoacetate Supplier & Distributor
- Available in bulk quantities for pharmaceutical and industrial use
- High-purity grades (98%, 99%, 99.5%)
- Technical and pharmaceutical grades available
- COA, MSDS, and specifications provided
MSDS
Includes detailed safety instructions, hazard identification, first aid measures, and storage guidelines.
Frequently Asked Questions (FAQs)
-
What is Ethyl Cyanoacetate used for?
Used as an activated methylene building block in pharmaceutical synthesis, Knoevenagel condensations, Michael additions, manufacturing heterocyclic compounds, and agrochemical synthesis. -
What is the chemical formula of Ethyl Cyanoacetate?
Molecular formula: C₅H₇NO₂ or NCCH₂COOC₂H₅ with molecular weight 113.12 g/mol. -
What is the structure of Ethyl Cyanoacetate?
Contains ethyl ester and nitrile group, both attached to the active methylene carbon; systematic name: ethyl 2-cyanoacetate.