Benzene | 71-43-2
Cas No :
71-43-2
Form :
Liquid
Molecular Weight :
78.11
Molecular Formula :
C6H6
Boiling Point :
80 °C
Melting Point :
5 °C
Flash Point :
10 °C
Solubility :
Water
Description :
Overview of Benzene
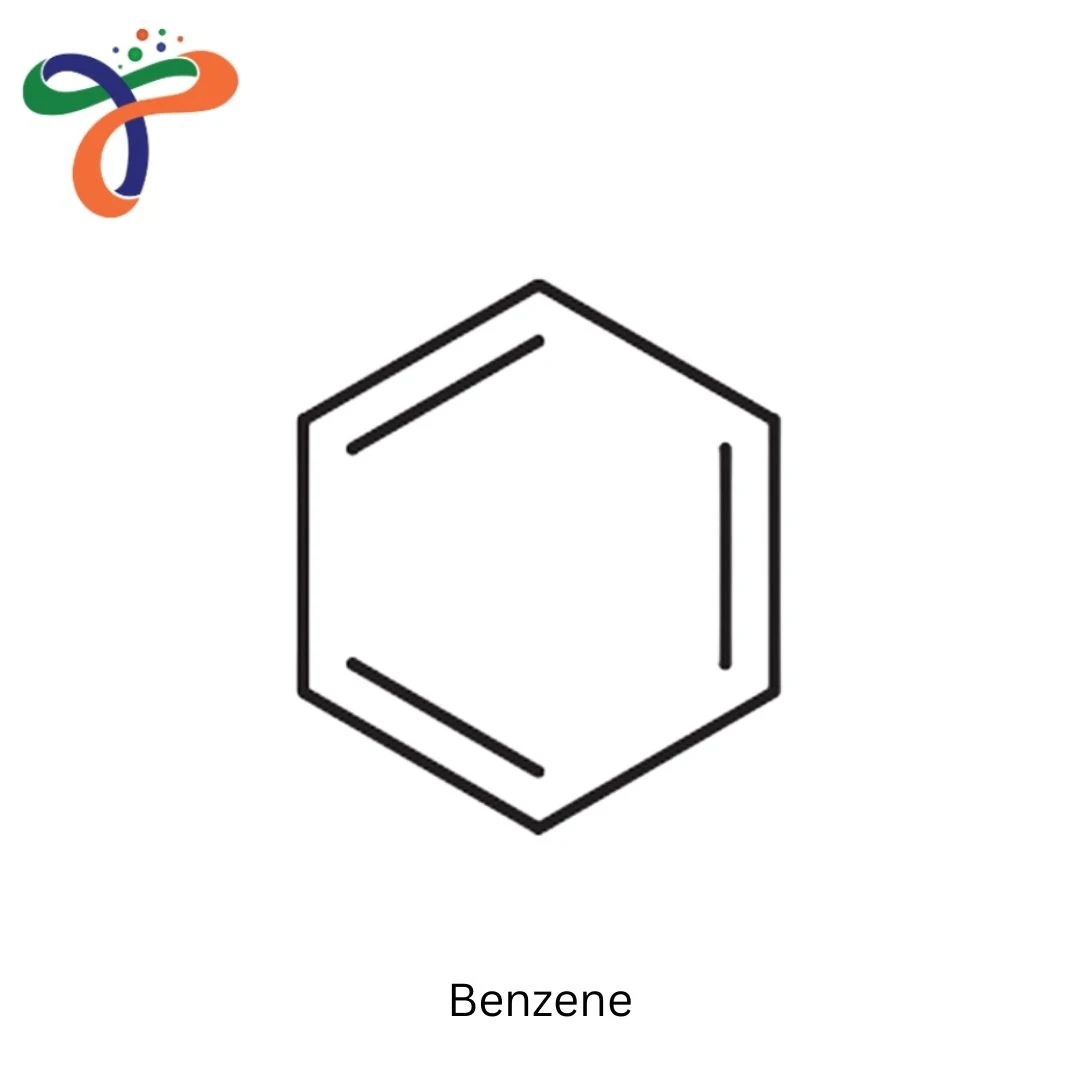
Benzene (C₆H₆) is a colorless, highly flammable liquid with a sweet odor, known for its extensive use as a starting material in the production of a wide range of chemicals. It is a simple aromatic hydrocarbon that consists of six carbon atoms bonded in a hexagonal ring, with alternating single and double bonds, forming a stable structure. Benzene is one of the most important industrial chemicals and is used in the production of plastics, synthetic fibers, and a variety of other chemical compounds.
The benzene formula is C₆H₆, and it is known for its distinctive hexagonal structure. As an aromatic compound, benzene exhibits a high degree of chemical stability, making it a valuable precursor in chemical synthesis.
Related products include Benzene Uses, Benzene Boiling Point, and Molecular Formula of Benzene.
Applications of Benzene
Chemical Manufacturing
- Benzene is widely used as a precursor in the production of various chemicals, including styrene, phenol, and aniline. These chemicals serve as building blocks for plastics, resins, and synthetic rubbers.
- It is also used in the production of polycarbonate plastics, which are commonly used in a variety of consumer products like eyeglasses, water bottles, and electronic components.
Solvent in Laboratories and Industry
- Benzene is used as a solvent in various industrial and laboratory applications due to its ability to dissolve a wide range of organic and inorganic compounds.
- It is used in the extraction of oils and fats, as well as in the formulation of paints, varnishes, and adhesives.
Pharmaceutical Industry
- Benzene is used as a precursor in the synthesis of drugs and pharmaceutical compounds. It is involved in the manufacture of various therapeutic agents and treatments.
- Benzene derivatives such as acetaminophen (paracetamol) are commonly used in pain relief formulations.
Aromatic Compounds Production
- Benzene is an essential raw material for the production of aromatic compounds used in the fragrance and flavor industries.
- It is involved in the manufacture of perfumes, flavoring agents, and other scented products.
Fuel Additives
- Benzene is used in the formulation of fuel additives to improve fuel performance in combustion engines.
- It is blended with gasoline to improve octane ratings, making it an important component in the fuel industry.
Safety & Handling Guidelines
- Store Benzene in a cool, dry, and well-ventilated area, away from heat, flames, and incompatible substances.
- Always handle Benzene in a fume hood or with proper ventilation due to its toxic and flammable nature.
- Use appropriate PPE, including gloves, goggles, and protective clothing when handling Benzene.
- Avoid inhalation of fumes and prevent skin or eye contact with the liquid.
- Benzene is a carcinogen, so it should be handled with extreme care to minimize exposure.
Where to Buy Benzene?
Benzene Manufacturer
ChemicalBull supplies high-quality Benzene for use in industrial applications, chemical manufacturing, and research, meeting international quality standards.
Benzene Supplier & Distributor
- Available in bulk for use in plastics, pharmaceuticals, solvents, and fuel additives
- Suitable for chemical synthesis, laboratory research, and industrial production
- COA, MSDS, and regulatory documents provided
MSDS
Includes detailed safety instructions, hazard identification, and storage guidelines.
Frequently Asked Questions (FAQs)
What is the formula of Benzene?
The formula of Benzene is C₆H₆, consisting of six carbon atoms bonded in a hexagonal ring with alternating single and double bonds.What is the molecular formula of Benzene?
The molecular formula of Benzene is also C₆H₆, which reflects its chemical composition as an aromatic hydrocarbon.What is the boiling point of Benzene?
The boiling point of Benzene is 80.1°C (176.2°F), making it a volatile compound that evaporates easily at room temperature.